Difference between revisions of "OSPAR eutrophication assessment"
(→Category I: Nutrient enrichment) |
|||
| Line 14: | Line 14: | ||
== Category I: Nutrient enrichment == | == Category I: Nutrient enrichment == | ||
| + | == Introduction == | ||
| + | |||
| + | The major causes of nutrient enrichment in coastal areas are associated with the need to satisfy human nutrition and diet by the use of fertilizers (anthropogenic nutrient loads). Nutrients in fertilizer may reach the estuary in several ways: through accidental release during production and transportation, by runoff from farm fields into surface or ground waters, or by incorporation into the food web via uptake by plants and consumption of plants by humans and livestock. Nutrients such as nitrates and phosphates are needed for the biomass production of phytoplankton and micro-and macrophytes (e.g. seagrasses, alges) and are the most common single factor causing eutrophication. | ||
| + | |||
| + | |||
| + | {|border="1" align=right cellspacing="0" cellpadding = "8" width="800px" | ||
| + | |+ '''<u>Table1: Nutrient enrichment'''</u> | ||
| + | |width="100px"| '''Nutrient concentration (µmol/l)''' | ||
| + | |width="50px"| '''Assessment''' | ||
| + | |- | ||
| + | | Ammonium<sup>1</sup> (NH<sub>4</sub><sup>+</sup>) | ||
| + | | Throughout the year | ||
| + | |- | ||
| + | | Nitrate<sup>2</sup> (NO<sub>3</sub><sup>-</sup>) | ||
| + | | Throughout the year | ||
| + | |- | ||
| + | | Nitrite<sup>3</sup> (NO<sub>2</sub><sup>-</sup>) | ||
| + | | Throughout the year | ||
| + | |- | ||
| + | | Phosphate (PO<sub>4</sub><sup>3-</sup>) | ||
| + | | Throughout the year | ||
| + | |- | ||
| + | | SiO4 (SiO<sub>4</sub><sup>4-</sup>) | ||
| + | | Throughout the year | ||
| + | |- | ||
| + | | Dissolved Inorganic Nitrogen (DIN) (1+2+3) | ||
| + | | Winter* | ||
| + | |- | ||
| + | | Dissolved Inorganic Phosphor (DIP) | ||
| + | | Winter* | ||
| + | |- | ||
| + | | Total Nitrogen (Includes all forms of nitrogen compounds) | ||
| + | | Calculated from all seasons | ||
| + | |- | ||
| + | | Total Phosphor (Includes all forms of phosphor compounds | ||
| + | | Calculated from all seasons | ||
| + | |- | ||
| + | |colspan ="2"| <small>*During phytoplankton blooms, inorganic nutrients in surface layers may be almost completely consumed, leading to nutrient limitation. This results in a large seasonal variability of nutrient concentrations. For this reason DIN and DIP are usually measured and assessed during winter, when biological activity is lowest (highest nutrient concentrations).</small> | ||
| + | |- | ||
| + | |} | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | === Nitrogen === | ||
| + | |||
| + | Nitrogen is used by living organisms to produce a number of complex organic molecules such as amino acids, proteins and nucleic acids. The store of nitrogen found in the atmosphere, where it exists mainly as a gas (mainly N<sub>2</sub>), plays an important role for life. Other major stores include organic matter in soil and the oceans. Nitrogen is often the most limiting nutrient for plant growth. This occurs because most plants can only take up nitrogen in two solid forms: ammonium ion (NH<sub>4</sub><sup>+</sup>) and the nitrate ion (NO<sub>3</sub><sup>-</sup>). Most plants obtain the nitrogen they need as inorganic nitrate from the soil solution. Ammonium is used less by plants for uptake because in large concentrations it is extremely toxic. The stimulation of plant growth by nitrates may result in eutrophication, especially due to algae. | ||
| + | |||
| + | === Phosphorus === | ||
| + | The primary biological importance of phosphates is as a component of nucleotides, which serve as energy storage within celles (ATP) or when linked together form the nucleic acids (DNA,RNA). Most of world's phosphorus is locked up in rocks and can only be released by weathering. A lot of the phosphorus that runs off into the ocean also gets buried into the ocean floor because it precipitates into solid form and settles to the bottom as sediment. Phosphorus is only available to organisms in small quantities. For this reason, phosphorus is a limiting factor for plant growth. Their presence in water is due to detergents, fertilizers and biological processes. An excess of phosphate will stimulate the growth of algae and aquatic plants. | ||
| + | |||
| + | === Silica === | ||
| + | Diatoms are the predominant siliceous organisms in the marine environment and are a major component of the phytoplankton community. Diatoms accumulate silica (SiO<sub>2</sub>) as a structural element in their cell walls. The silica need of a diatom may be so great relative to the silica concentration of seawater, that it restricts diatom growth. | ||
| + | |||
| + | == Measuring devices == | ||
| + | * [[Nutrient analysers|Nutrient analysers]] | ||
| + | |||
| + | == References == | ||
| + | #OSPAR (2005), Common Procedure for the Identification of the Eutrophication Status of the OSPAR Maritime Area (Reference number: 2005-3).EUC 2005 Summary Record – EUC 05/13/1, Annex 5 as amended and endorsed by OSPAR 2005 Summary Record - OSPAR 05/21/1, §§ 6.2-6.5 and Annex 6. | ||
| + | #HELCOM (2009), Eutrophication in the Baltic Sea – An integrated thematic assessment of the effects of nutrient enrichment and eutrophication in the Baltic Sea region. Balt. Sea Environ. Proc. No. 115B. | ||
| + | #Jesper H. Andersen, Louise Schlüter and Gunni Aertebjerg (2006). Coastal eutrophication: recent developments in definitions and implications for monitoring strategies. ''Journal of Plankton Research''. '''28 (7)''', 621-628. | ||
== Category II: Nutrient enrichment == | == Category II: Nutrient enrichment == | ||
Revision as of 12:50, 18 February 2013
Coastal eutrophication is a growing marine environmental problem in Europe and the need for more effective monitoring and control measures is increasing. The OSPAR Commission adopted the Common Procedure for the Identification of the Eutrophication Status of the maritime area in 1997. OSPAR committed all members to reduce phosphorus and nitrogen inputs into the maritime areas and 'to combat eutrophication to achieve a healthy environment where eutrophication does not occur'.
The Common Procedure is used to assess the eutrophication status of the OSPAR maritime area in a harmonized way. This is based on classifications in terms of the following types of areas: Non-problem Areas, Potential Problem Areas and Problem Areas.
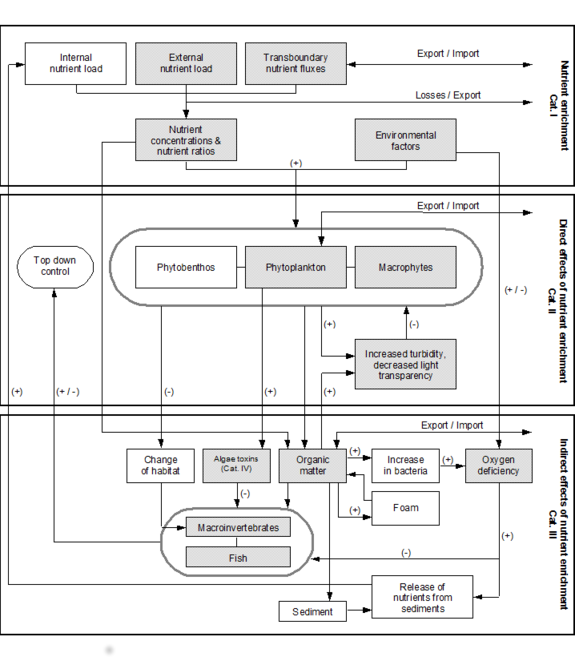
The Common Procedure includes the main parameters involved in eutrophication processess which are roughly differentiated in four categories (see figure)
- Category I: Nutrient enrichment
- Category II: Direct effects of nutrient enrichment
- Category III: Indirect effects of nutrient enrichment
- Category IV: Other effects of nutrient enrichment
Contents
Category I: Nutrient enrichment
Introduction
The major causes of nutrient enrichment in coastal areas are associated with the need to satisfy human nutrition and diet by the use of fertilizers (anthropogenic nutrient loads). Nutrients in fertilizer may reach the estuary in several ways: through accidental release during production and transportation, by runoff from farm fields into surface or ground waters, or by incorporation into the food web via uptake by plants and consumption of plants by humans and livestock. Nutrients such as nitrates and phosphates are needed for the biomass production of phytoplankton and micro-and macrophytes (e.g. seagrasses, alges) and are the most common single factor causing eutrophication.
| Nutrient concentration (µmol/l) | Assessment |
| Ammonium1 (NH4+) | Throughout the year |
| Nitrate2 (NO3-) | Throughout the year |
| Nitrite3 (NO2-) | Throughout the year |
| Phosphate (PO43-) | Throughout the year |
| SiO4 (SiO44-) | Throughout the year |
| Dissolved Inorganic Nitrogen (DIN) (1+2+3) | Winter* |
| Dissolved Inorganic Phosphor (DIP) | Winter* |
| Total Nitrogen (Includes all forms of nitrogen compounds) | Calculated from all seasons |
| Total Phosphor (Includes all forms of phosphor compounds | Calculated from all seasons |
| *During phytoplankton blooms, inorganic nutrients in surface layers may be almost completely consumed, leading to nutrient limitation. This results in a large seasonal variability of nutrient concentrations. For this reason DIN and DIP are usually measured and assessed during winter, when biological activity is lowest (highest nutrient concentrations). | |
Nitrogen
Nitrogen is used by living organisms to produce a number of complex organic molecules such as amino acids, proteins and nucleic acids. The store of nitrogen found in the atmosphere, where it exists mainly as a gas (mainly N2), plays an important role for life. Other major stores include organic matter in soil and the oceans. Nitrogen is often the most limiting nutrient for plant growth. This occurs because most plants can only take up nitrogen in two solid forms: ammonium ion (NH4+) and the nitrate ion (NO3-). Most plants obtain the nitrogen they need as inorganic nitrate from the soil solution. Ammonium is used less by plants for uptake because in large concentrations it is extremely toxic. The stimulation of plant growth by nitrates may result in eutrophication, especially due to algae.
Phosphorus
The primary biological importance of phosphates is as a component of nucleotides, which serve as energy storage within celles (ATP) or when linked together form the nucleic acids (DNA,RNA). Most of world's phosphorus is locked up in rocks and can only be released by weathering. A lot of the phosphorus that runs off into the ocean also gets buried into the ocean floor because it precipitates into solid form and settles to the bottom as sediment. Phosphorus is only available to organisms in small quantities. For this reason, phosphorus is a limiting factor for plant growth. Their presence in water is due to detergents, fertilizers and biological processes. An excess of phosphate will stimulate the growth of algae and aquatic plants.
Silica
Diatoms are the predominant siliceous organisms in the marine environment and are a major component of the phytoplankton community. Diatoms accumulate silica (SiO2) as a structural element in their cell walls. The silica need of a diatom may be so great relative to the silica concentration of seawater, that it restricts diatom growth.
Measuring devices
References
- OSPAR (2005), Common Procedure for the Identification of the Eutrophication Status of the OSPAR Maritime Area (Reference number: 2005-3).EUC 2005 Summary Record – EUC 05/13/1, Annex 5 as amended and endorsed by OSPAR 2005 Summary Record - OSPAR 05/21/1, §§ 6.2-6.5 and Annex 6.
- HELCOM (2009), Eutrophication in the Baltic Sea – An integrated thematic assessment of the effects of nutrient enrichment and eutrophication in the Baltic Sea region. Balt. Sea Environ. Proc. No. 115B.
- Jesper H. Andersen, Louise Schlüter and Gunni Aertebjerg (2006). Coastal eutrophication: recent developments in definitions and implications for monitoring strategies. Journal of Plankton Research. 28 (7), 621-628.
Category II: Nutrient enrichment
Category III: Nutrient enrichment
References
- OSPAR (2005), Agreement on the Eutrophication Monitoring Programme(Reference number: 2005-4).EUC 2005 Summary Record – EUC 05/13/1, Annex 6.
- Ulrich Claussen, Wanda Zevenboom, Uwe Brockmann, Dilek Topcu and Peter Bot (2009). Assessment of the eutrophication status of transitional, coastal and marine waters within OSPAR. Hydrobiologia. 629, 49-58.