Biosensing
The development and use of high resolution biosensing techniques allow us to monitor the in situ marine environment.[1] The monitoring can vary between the analysis of the water quality, prediction and detection of Harmful Algal Blooms (HAB) and the estimation of environmental and human health risks. Examples of tools based on automated sensing techniques to monitor environmental microbial related variables are listed below. [1]
Fig 1. Harmful Algal Blooms (HAB).[2]
Microalgal fiber optic biosensors[3]
Microalgal fiber optic biosensors are used for the detection of pesticides in (marine) water. The technique supports upon the photosynthetic properties of Chlorophyceae microalgae. These organisms are immobilized into a porous silicon layer that acts as a recognition element. The pesticide (simarine) enables the photosynthesis process of the microalgae and consequently O2 production. When the device comes in contact with water, the O2 production is monitored. The fluctuations or the absence of O2 are indications of the presence of pesticides.
Fig 2. Chlorophyceae .[4]
Automated online optical biosensing systems (AOBS) [5]
One of the consequences of global warming is the rise of cyanobacterial blooms. Cyanobacterial blooms generally proceed with the production of cyanotoxins that can be harmful for humans. Online optical biosensing systems are developed for the rapid detection of microcystin-LR (MC-LR), a harmful toxic substance. AOBS consists partly of Molecular MC-LR-ovalbumin that is captured/immobilized onto the surface of biochips. When AOBS comes in contact with high concentrations of MC-LR the fluorescence labeled antibodies bound on to the biochip surface will give a lower fluorescence signal. The intensity of the fluorescence signal is monitored to detect the different concentrations of toxins present in the water sample. This technique has already been successfully tested in the field.
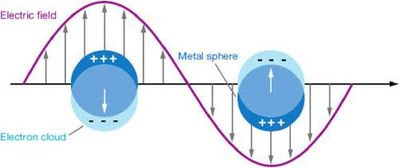
Surface plasma resonance (SPR) [1][6]
Surface Plasmon Resonance is a phenomenon associated with the coupling of light and collective oscillation of electrons at a metal-dielectric interface. [7] These oscillations are referred to as surface plasmon waves and their confined electromagnetic fields to the characterization of binding interactions of biomolecules on metallic surfaces. [6] This phenomenon has an interesting application for usage as a biosensor. The water sample will be brought in contact with the metallic sensor surface. This surface contains biomolecular recognition elements. The specific binding of the target molecules to the recognition elements is monitored and these bindings result in local refractive index variations that can be observed as changes in the spR reflectivity signal. [8]
Fig 3. The phenomenon of surface plasma resonance (SPR).[9]
Wearable electrochemical sensors[10]
This technique helps to detect heavy metal contaminants and nitroanaromatic explosives in seawater. The sensors that are able to detect the presence of these pollutants are implemented in the synthetic rubber neoprene. They form wearable screen printed electrochemical sensors that can for example be implemented in wet and dry suits. In this way data and warnings can be collected in an easy way by Oceanographers, local water-quality agencies and even recreational surfers/divers.
Biosensors to detect marine toxins in seafood[11]
This subject is not directly environmental based but is worth mentioning. More and more toxins are piling up in our food and shellfish for example accumulation of phytotoxin in their digestive glands. These toxins are harmless for shellfish, but toxic for humans. DSP (Diarrheic Shellfish Poisoning), PSP (Paralytic Shellfish Poisoning) are only a few of the diseases that are correlated with phytotoxins. There is an increasing demand for cheap and correct screening methods to detect these toxins. Biosensors have the potential to fulfill this demands. Examples of such biosensors are Sodium channel based hormones[12][13] Immune sensors[14][15][16] , enzyme inhibition-based biosensors[17], molecularly imprinted polymers (MIP-based sensor) [18] and finally chemosensors. [19][20][21]
Fig 4. A Paralytic Shellfish Poisoning warning sign. [22]
References
- ↑ 1.0 1.1 1.2 Querellou, J. (2010) MarineBiotechnology: A new vision and strategy for Europe. Marine Board-ESF Position Paper 15.
- ↑ http://www.oceanchampions.org
- ↑ Orellana, G., Villen, L., Haigh, D. (2007) Microalgal fiber-optic biosensors for water quality monitoring. Proceeding of society of photo-optical instrumentation engineers (spie), 6619: 61921-61921
- ↑ http://www.examplesof.net/2014/04/example-of-green-algae-chlorophyceae.html#.VZzRaLfvqUk
- ↑ Han-Chang, S. et al. (2013). Automated online optical biosensing system for continuous real-time determination of microcystin-LR with high sensitivity and specificity: early warning for cyanotoxin risk in drinking sources. Environ. Sci. Technol. 47: 4434-441
- ↑ 6.0 6.1 Méjard, R., Dostalek, J., Huang, C., Griesser, H. and Thierry, B. (2013). Tuneable and robust long range surface plasmon resonance for biosensing applications. Optical Materials 35: 2507-2513
- ↑ Raether, H. (1988). Surface plasmons on smooth and rough surfaces and on gratings. Springer Verlag, Berlin
- ↑ Homola, J. (2008). Surface plasmons resonance sensors for detection of chemical and biological species. Chem. Rev. 108(2): 462-493
- ↑ https://gregemmerich.wordpress.com/2012/11/16/surface-plasmon-resonance-technology-overview-and-practical-applications/
- ↑ Malzahn, K., Windmiller, J. R., Valdes-Ramirez, G., Schoning, M. J. and Wang, J. (2011). Wearable electrochemical sensors for in situ analysis in marine environments. Analyst, 136: 2912-2917
- ↑ Campas, M., Prieto-Simon, B. and Marty, J. (2007). Biosensors to detect marine toxins: Assessing seafood safety. Talanta 72: 884-895
- ↑ Cheun, B., Endo, H., Hayashi, T., Nagashima, Y. and Watanabe, E. (1996). Biosens. Bioelectron. 11: 1185.
- ↑ Cheun, B., Loughran, M., Hayashi, T., Nagashima, Y. and Watanabe, E. (1998). Toxicon 36: 1371
- ↑ Tang, A.X.J., Pravda, M., Guilbault, G. G., Pilestky, S. and Turner, A. P. F. (2002). Anal. Chim. Acta 471: 33
- ↑ Marquette, C. A., Coulet, P. R. and Blum, L. J. (1999). Anal. Chim.. Acta 398: 173
- ↑ Chen, Q. Yu, S., Taylor, A. D., Homola, J., Hock, B. and Jiang, S. (2005). Sens. Actuator B 107: 193
- ↑ Hamada-Sato, N., Minamitani, N., Inaba, Y., Nagashima, Y., Kobayashi, T., Imada, C. and Watanabe, E. (2004). Sensor Mater. 16: 99
- ↑ Lotierzo, M., Henry, O.Y.F., Piletsky, S., Tothill, I., Cullen, D., Kania, M., Hock, B. and Turner, A.P.F. (2004). Biosens. Bioelectron. 20: 145
- ↑ Gawley, R. E., Pinet, S., Cardona, C.M., Datta, P. K., Ren, T., Guida, W. C., Nydick, J. and Leblanc, R. M.(2002). Am. Chem. Soc. 124: 13448
- ↑ Kele, P., Orbulescu, J., Calhoun, T. L., Gawley, R. E. and Leblanc, R. M. (2002). Tetrahedron Lett. 43: 4413
- ↑ Gawley, R. E., Shanmugasundaram, M., Thorne, J.B. and Tarkka, R. M. (2005). Toxicon 45: 783
- ↑ http://globalbiodefense.com/2013/08/08/government-seeks-field-detectors-for-brevetoxins/