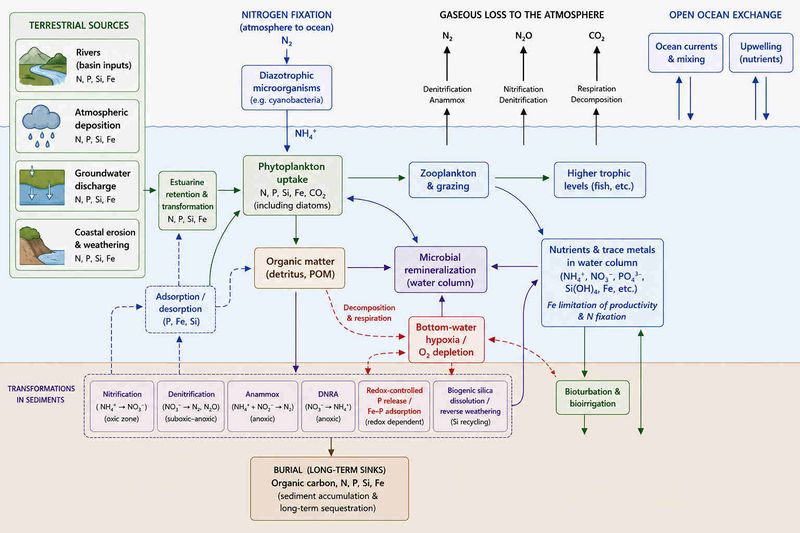
Nutrient conversion in the marine environment
Coastal and shelf seas play a central role in global biogeochemical cycles by supporting high levels of primary production and intense recycling of nutrients and organic matter. Marine phytoplankton, together with bacteria and other marine microorganisms, convert dissolved nutrients and carbon dioxide into organic matter that sustains marine food webs and contributes to the global Ocean carbon sink. Nutrient availability therefore strongly influences marine ecosystem productivity, carbon sequestration and oxygen dynamics.
Human activities have greatly increased the delivery of nitrogen and phosphorus to coastal waters through agriculture, wastewater discharge and atmospheric deposition, leading to widespread eutrophication. Excess nutrient enrichment stimulates algal growth and plankton blooms, which can alter food webs, increase oxygen consumption during decomposition and contribute to coastal hypoxia, acidification and biodiversity loss (see Possible consequences of eutrophication and Ocean acidification).
Coastal ecosystems also play an important role in climate regulation. Part of the organic carbon produced in coastal waters is buried in sediments or stored in vegetated coastal habitats such as salt marshes, mangroves and seagrass meadows, contributing to Blue carbon sequestration. Climate change is expected to modify nutrient cycling and marine productivity through ocean warming, stronger stratification, deoxygenation and changing riverine nutrient inputs, with important feedbacks on the global carbon cycle and coastal ecosystem functioning.
This article first discusses the main natural and anthropogenic sources of nutrients to the marine environment. It then describes the general processes of nutrient cycling and organic matter remineralization in coastal and shelf seas, with particular emphasis on the role of sediments and microbial activity. Subsequent sections examine the transformation pathways of the major nutrients nitrogen, phosphorus and silicon, including processes such as nitrification, denitrification, phosphorus recycling and silica dissolution. The final section briefly discusses the role of trace metals in marine biogeochemical cycles and their influence on marine productivity and nutrient conversion. Major pathways and processes involved in biogeochemical cycles in the coastal marine environment are schematically represented in Fig. 1.
Contents
Nutrient sources
Nutrients in coastal environments originate from multiple sources, including rivers, atmospheric deposition, groundwater discharge, and in situ biological fixation. Rivers are the primary transport pathway of nutrients to coastal oceans. Riverine inputs of nitrogen (N), mainly as nitrate (NO3−), ammonia (NH3) and ammonium (NH4+), and phosphorus (P), mainly as orthophosphate (PO43−), approximately doubled between 1960 and 1990[1]. This increase is largely attributable to the expanded use of synthetic fertilizers in agriculture. Additional anthropogenic sources include wastewater discharge from urban sewer systems[2] and industrial effluents (see also What causes eutrophication?). Natural processes such as rock weathering[3] and terrestrial nitrogen fixation[4] also contribute to riverine nitrogen export.
Around 2015, global riverine nitrogen discharge to coastal waters was estimated at 35–50 Tg N yr−1[5][6][7][8][9]. Riverine discharge of reactive phosphorus is estimated at 2–5 Tg P yr−1[10][1][5][9] (1 Tg = 1012 g).
Submarine groundwater discharge may contribute nutrient fluxes comparable to or exceeding riverine inputs globally, although estimates remain uncertain[11]. Atmospheric deposition contributes approximately 8 Tg N yr−1 to continental shelves and 40–50 Tg reactive N yr−1 to the global ocean[6]. Regional differences are substantial. For example, atmospheric deposition accounts for about 30% of total land-based nitrogen inputs to the North Sea and about 50% to the Baltic Sea[12]. The N:P ratio of atmospheric deposition can be very high; in the North Sea it was estimated at 503:1, far exceeding the Redfield ratio of 16:1 typical of phytoplankton[13].
In addition to external inputs from land, bioavailable nitrogen is produced in situ through biological nitrogen fixation (diazotrophy). Specialized diazotrophic cyanobacteria, such as the photoautotroph Trichodesmium and symbiotic unicellular cyanobacteria (e.g. UCYN-A), convert dissolved dinitrogen gas (N2) into ammonium (NH4+) using the enzyme nitrogenase[14][15]. This process occurs predominantly in warm, well-lit, oligotrophic ocean regions. Global marine nitrogen fixation is estimated at 70–170 Tg N yr−1[16][17][18]. Additional nitrogen fixation occurs in aphotic ocean regions by bacteria and archaea, contributing an estimated 13–134 Tg N yr−1[19]. Coastal and benthic nitrogen fixation adds approximately 15 Tg N yr−1[17].
In addition to nitrogen and phosphorus, silicon (Si) is an essential nutrient for silicifying organisms such as diatoms, silicoflagellates, certain radiolarians, rhizarians, and siliceous sponges. Dissolved Si (dSi), mainly as undissociated monomeric silicic acid, Si(OH)4, is the only Si compound available for uptake by marine organisms. Rivers are the main source of dissolved silicon to the ocean, followed by submarine groundwater, mineral dissolution, hydrothermal inputs, aeolian dust, and glacial weathering. A large reservoir of dissolved silicon exists in the deep ocean, which becomes available to surface ecosystems through upwelling[20].
Nutrient cycling
Nutrient cycling is a process in which marine microorganisms play a crucial role. The key steps include the uptake of nutrients by phytoplankton to produce organic matter and the subsequent release of these nutrients during respiration and mineralization of organic matter by bacteria, making them available again for biological uptake (see Fig. 1 and Plankton bloom). Oxygen availability is a major regulator of marine nutrient cycling because it controls the balance between aerobic and anaerobic microbial pathways, influences sediment–water nutrient exchange, and governs the retention or removal of nutrients from marine ecosystems.
Aerobic water column remineralization of organic matter into carbon dioxide, nitrate and phosphate is commonly represented by[21]
[math]C_{106}H_{175}O_{42}N_{16}P + 150 O_2 \rightarrow 106 CO_2 + 16 HNO_3 + H_3PO_4 + 78 H_2O[/math].
Coastal shelf seas are zones of intense nutrient cycling, which enhances primary productivity[22]. Shelf seas are estimated to account for up to 80% of global benthic mineralization, despite covering only about 7% of the seafloor[23]. Most mineralization occurs after detrital plankton settles on the seabed and is decomposed under aerobic or anaerobic conditions.
In coarse-grained sediments, organic matter is rapidly mineralized because oxygen penetrates deeply into the sediment due to its high permeability[24]. As a result, oxygen consumption rates are high and organic carbon storage is low[25]. Mineralization is further enhanced by bioturbating benthic macrofauna, which increase sediment mixing and oxygen penetration[26] (Fig. 2).
In fine-grained cohesive sediments, oxygen penetration is limited; anaerobic mineralization pathways therefore become relatively more important, and organic carbon accumulates to higher levels[27].
In deeper parts of the continental shelf and the open ocean, less organic material reaches the seabed because a larger fraction is decomposed in the water column before settling[28]. See also Ocean carbon sink.
Nutrient transformation
Nitrogen
Nitrogen (N) species in aquatic environments include dissolved inorganic forms (nitrate NO3−, nitrite NO2−, ammonium NH4+), dissolved organic nitrogen, and particulate organic nitrogen[29]. Nitrogen is removed from aquatic systems by sedimentation and burial, and most importantly by conversion to atmospheric gases through denitrification coupled to organic matter decomposition (see Fig. 1). The decomposition of organic matter by bacteria and fungi yields primarily ammonium (NH4+) rather than nitrate (NO3-) as it is released directly from organic nitrogen without requiring an additional microbial oxidation step.
Denitrification is the microbial reduction of nitrate to gaseous nitrogen species, mainly dinitrogen (N2) and nitrous oxide (N2O), during the oxidation of organic matter. Denitrification generally depends on prior nitrification, in which ammonium is oxidized to nitrite and nitrate by autotrophic bacteria and archaea under aerobic conditions. These microorganisms are chemoautotrophs because they obtain energy from the oxidation of inorganic nitrogen compounds rather than from sunlight. Ammonium oxidizers are bacteria such as Nitrosomonas spp, and nitrite oxidizers are bacteria such as Nitrobacter spp.The different nitrification steps can be represented by
Ammonification: [math]\text{org.}N \rightleftharpoons NH_4^+[/math] => ammonium oxidation: [math]2NH_4^+ + 3O_2\rightarrow 2NO_2^- + 4H^+ + 2H_2O[/math] => nitrite oxidation: [math]2NO_2^- +O_2 \rightarrow 2NO_3^-[/math].
In the presence of comammox bacteria, ammonium can be directly oxidized to nitrate (COMplete AMMonia OXidation).
Denitrification is a heterotrophic process that occurs under low-oxygen conditions (typically ≤0.2 mg O2 L−1)[30][22]. Denitrifying bacteria are widespread; the main controlling factors are nitrate or nitrite availability, low oxygen concentrations, and the presence of labile organic carbon. This organic carbon, including extracellular polymeric substances (EPS) produced by marine microorganisms, serves as an electron donor in the denitrification process.
Because nitrification requires oxygen and denitrification requires low oxygen conditions, their coupling occurs primarily in environments with sharp oxygen gradients. Muddy shelf-sea sediments provide such conditions[31][30]. Nitrate produced in the aerobic surface layer diffuses into deeper suboxic layers, where denitrification takes place.
Nitrogen can also be removed in the absence of organic matter through anaerobic ammonium oxidation. This microbial process, known as anammox, converts ammonium and nitrite directly into dinitrogen gas (N2) and water under anoxic conditions[32]. Biological activity of benthic macrofauna can substantially extend the depth of the oxic–suboxic interface through bioturbation and bioirrigation, thereby enhancing coupled nitrification–denitrification and increasing overall nitrogen removal[33].
Denitrifying bacteria compete with microorganisms performing dissimilatory nitrate reduction to ammonium (DNRA) [34]. Unlike denitrification, DNRA retains nitrogen in the ecosystem by converting nitrate into ammonium, which remains available for biological uptake. DNRA is common in low-oxygen environments such as hypoxic sediments and oxygen minimum zones. Ammonium produced by DNRA can subsequently be converted to dinitrogen gas through anammox[35].
Globally, most denitrification occurs in shelf-sea sediments (Fig. 3), where nitrogen removal is estimated at 200–300 Tg N yr−1[30]. Additional denitrification occurs in oxygen-deficient regions of the open ocean, particularly in the Eastern Tropical North Pacific, Eastern Tropical South Pacific, and the Arabian Sea, where approximately 50–100 Tg N yr−1 is removed[36][30].
Denitrification also occurs in estuaries. Significant denitrification has been observed where a strong estuarine turbidity maximum is present[37]; it is surmised that aerobic degradation of suspended organic particles creates hypoxic microniches for the growth of denitrifying bacteria. Important denitrification has also been reported in benthic intertidal ecosystems, both in the freshwater and salt water tidal reaches[38], with microalgae potentially playing an important role[39]. A field study in a tropical estuary (Northern Australia) revealed intertidal mudflats per unit area to be hotspots of N removal, with higher denitrification efficiency than other benthic habitats, including mangrove and saltmarsh[40].
Denitrification rates generally increase with temperature, partly because warming promotes oxygen depletion and enhances microbial activity[41]. Denitrifying microbial communities are also sensitive to salinity variations, which can influence nitrogen removal efficiency in coastal systems[42].
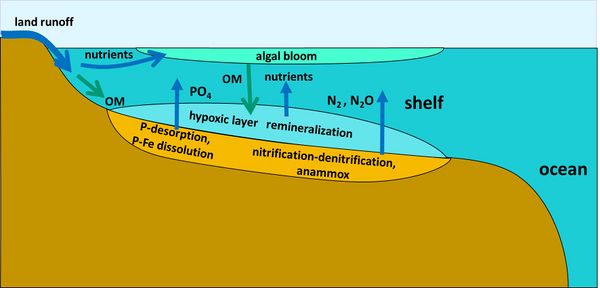
Phosphorus
Phosphorus (P) discharged by rivers into coastal waters occurs in dissolved and particulate forms, including dissolved inorganic phosphorus (DIP), dissolved organic phosphorus (DOP), particulate inorganic phosphorus (PIP), and particulate organic phosphorus (POP). Much of the non-reactive PIP, such as apatite, is deposited on continental shelves and does not reach the open ocean[44].
A significant fraction of riverine phosphate is adsorbed onto clay particles and associated with iron and manganese oxides or oxyhydroxides. These particles are often retained in estuaries, but increasing salinity promotes desorption, releasing phosphate into coastal waters[45][46]. It has been estimated that the amount of phosphate released through desorption from suspended particles may be two to five times greater than the dissolved phosphate directly delivered by rivers[44].
Hypoxic conditions in coastal waters promote the release of dissolved inorganic phosphorus from sediments. Under low-oxygen conditions, iron oxides that bind phosphate are reduced, releasing phosphate into pore water and subsequently into the overlying water column[47]. This internal phosphorus recycling can sustain or enhance algal blooms (Fig. 3).
Particulate phosphorus deposited in sediments may also be released into pore waters during diagenesis, contributing to benthic phosphorus fluxes to bottom waters[44]. Dissolved inorganic phosphorus, mainly in the form of orthophosphate (PO43−), is assimilated by phytoplankton and incorporated into organic matter, forming DOP and POP. This organic phosphorus is returned to the dissolved inorganic pool through microbial mineralization during decomposition. Most DOP is mineralized in surface waters, whereas the fraction transported to deeper ocean layers may persist for thousands of years due to slow turnover rates. Permanent removal of phosphorus from the ocean occurs primarily through burial in marine sediments, estimated at approximately 3–10 Tg P yr−1[48].
Phosphorus may also be removed through microbial reduction of phosphate to gaseous phosphine (PH3), although this pathway is poorly quantified and its global significance remains uncertain[49][29].
Silicon
Silicon (Si) in the marine environment occurs mainly as dissolved silicon (dSi), primarily in the form of silicic acid Si(OH)4, and as particulate silicon in the form of biogenic silica (bSiO2, also called opal). Biogenic silica consists of amorphous silica contained in living organisms and in biogenic detritus in the water column, soils, and sediments. The main transformation processes are the biological uptake of dissolved silicon and its biomineralization into biogenic silica, and the subsequent dissolution of biogenic silica back into dissolved silicon[2].
The silicon cycle differs from other nutrient cycles in several ways:[21]
- silicic acid is utilized almost exclusively by diatoms, whereas nitrate and phosphate are utilized by all phytoplankton;
- siliceous material is not passed up the food chain to a significant degree;
- regeneration of opal to silicic acid is due to dissolution, not metabolic degradation;
- Si exists in seawater only in its inorganic form, primarily as silicic acid, whereas N and P (especially N) exist in several inorganic and organic forms.
Over long time scales, biogenic silica undergoes chemical and mineralogical transformations during diagenesis[50]. These transformations may include conversion of opaline silica into aluminosilicate minerals such as clays[51] (often designated as authigenic aluminosilicate minerals as they are formed where observed). Aluminosilicate minerals have a much lower dissolution rate than opal and therefore promote Si burial in the sediment bed.
Diatoms are the dominant producers of biogenic silica in the marine environment. Radiolarians, siliceous sponges, and chrysophytes can also contribute locally to biogenic silica production[52]. Global marine biogenic silica production is estimated at 7000–8000 Tg Si yr−1[2]. Most biogenic silica produced in surface waters dissolves in the upper ocean or during sinking, with only about one third reaching the seafloor[53]. Much of the deposited silica is recycled at the sediment–water interface, and only a small fraction, approximately 250 Tg Si yr−1, is ultimately buried in marine sediments. The total oceanic dissolved silicon inventory is estimated at about 3.36 × 106 Tg Si, most of which resides in deep ocean waters, particularly in the Pacific and Southern Oceans[21].
The primary external source of dissolved silicon to the ocean is river discharge, derived from continental weathering. Total dissolved silicon input to the ocean, including contributions from groundwater, hydrothermal systems, and atmospheric dust, is estimated at 340–490 Tg Si yr−1[2]. Losses from the marine silicon pool occur mainly through sediment burial and reverse weathering, in which dissolved or biogenic silica is incorporated into authigenic aluminosilicate minerals, as well as through accumulation in siliceous sponge skeletons[2].
Although these fluxes are small relative to the large oceanic dissolved silicon reservoir, regional silicon availability is sensitive to changes in supply and removal. Human activities such as dam construction reduce silicon delivery to coastal waters by trapping biogenic and particulate silica in reservoirs. This effect has already altered silicon cycling in coastal zones downstream of major dammed rivers such as the Nile and Danube[20]. Reduced silicon availability and associated changes in nutrient stoichiometry, particularly the N:P:Si ratios, can shift phytoplankton communities from diatom-dominated systems toward non-siliceous algae and cyanobacteria. Important impacts on food-web structure and oxygen dynamics have been observed, potentially promoting harmful algal blooms and altering carbon export[54].
Climate change is also expected to influence the marine silicon cycle. Increased stratification of the ocean surface reduces vertical transport of dissolved silicon from deep waters, potentially limiting diatom growth. Conversely, melting sea ice in polar regions may locally increase dissolved silicon availability. Model projections suggest a global decline in diatom biomass over the coming century, except in parts of the Southern Ocean[55][56].
Trace metals
Very small concentrations of dissolved metals, known as trace metals (including iron Fe, manganese Mn, zinc Zn, copper Cu, cobalt Co, nickel Ni, and cadmium Cd), play essential roles in marine biogeochemical cycles. Trace metals are supplied to continental shelves through rivers and estuaries, where a substantial fraction is removed by adsorption onto fine sediment particles. In contrast, iron and manganese are delivered to the open ocean primarily through atmospheric deposition of mineral dust. Additional sources include hydrothermal vents and sediment release.[57]
Trace metals occur in various chemical forms, and their biological availability is regulated by photochemical reactions and microbial processes that modify their oxidation state and complexation with organic ligands[57]. Rapid recycling of trace metals takes place in surface waters, where dissolved concentrations are extremely low due to efficient biological uptake and regeneration.
Trace metals are essential components of metalloenzymes that regulate key metabolic processes and serve structural roles in proteins[57]. Although required only in trace amounts, both deficiency and excess can be harmful: insufficient availability limits biological productivity, while elevated concentrations may be toxic. Marine organisms have evolved to function within narrow trace metal concentration ranges, making marine ecosystems sensitive to environmental changes that alter trace metal availability. Ocean acidification may influence trace metal speciation and bioavailability, although the mechanisms remain incompletely understood (see Ocean acidification#Bioavailability of trace metals).
Trace metals strongly influence primary production and nutrient cycling. For example, phytoplankton growth can be limited by iron availability in large ocean regions. Iron is also essential for nitrogen fixation by diazotrophic cyanobacteria such as Trichodesmium. Limited trace metal availability can therefore constrain the marine nitrogen cycle, since many nitrogen transformation processes rely on metalloenzymes containing iron, copper, or molybdenum. Trace metals are also involved in carbon fixation (requiring Fe and Mn), organic matter remineralization (Fe, Zn), methane oxidation (Cu), calcification (Zn, Co), and silica biomineralization in some organisms (Zn, Cd, Se)[57].
Synthesis and environmental significance
Nutrient cycling in coastal and shelf seas is governed by tightly coupled biological, chemical and physical processes that regulate the transformation, recycling, retention and removal of nitrogen, phosphorus, silicon and trace metals. Marine microorganisms play a central role in these cycles by mediating primary production, organic matter remineralization and redox-dependent nutrient transformations. Oxygen availability strongly influences the balance between nutrient recycling and permanent nutrient removal, particularly in coastal sediments where nitrification, denitrification, anammox and phosphorus release occur in close spatial association.
Human activities have substantially altered marine nutrient cycles by increasing nutrient inputs from agriculture, wastewater discharge and atmospheric deposition, while climate change is modifying ocean stratification, circulation, oxygen concentrations and nutrient transport. These changes affect nutrient stoichiometry and can shift phytoplankton communities from diatom-dominated systems toward non-siliceous algae and cyanobacteria, with important consequences for food webs, fisheries productivity, carbon sequestration and ecosystem health. Coastal eutrophication, hypoxia and ocean acidification are therefore closely interconnected consequences of disturbances in marine biogeochemical cycling.
Continental shelf sediments act both as reactors that recycle nutrients back to the water column and as sinks where nutrients and organic carbon are permanently buried or removed through gaseous pathways. Understanding the interactions between nutrient cycling, climate forcing and ecosystem functioning is essential for predicting future changes in marine productivity and for developing effective strategies to reduce eutrophication and protect coastal ecosystems.
Appendix: Estimates of major natural and anthropogenic nutrient sources and sinks
Nitrogen (N)
Major sources of marine nitrogen
| Source | Type | Approximate flux | Main references |
|---|---|---|---|
| River discharge | Mixed natural + anthropogenic | 35–50 Tg N yr−1 | [5] [6] [9] |
| Atmospheric deposition | Mainly anthropogenic | 40–50 Tg reactive N yr−1 to the global ocean; ~8 Tg N yr−1 to continental shelves | [6] [58] |
| Marine biological nitrogen fixation | Natural | 70–170 Tg N yr−1 | [16] [17] [59] |
| Deep-ocean nitrogen fixation | Natural | 13–134 Tg N yr−1 | [60] |
| Coastal and benthic nitrogen fixation | Natural | ~15 Tg N yr−1 | [17] |
| Submarine groundwater discharge | Mixed | Poorly constrained; regionally important | [61] |
Major sinks of marine nitrogen
| Sink | Type | Approximate flux | Main references |
|---|---|---|---|
| Shelf-sea denitrification | Natural microbial sink | 200–300 Tg N yr−1 | [30] |
| Open-ocean denitrification | Natural | 50–100 Tg N yr−1 | [36][30] |
| Anammox | Natural microbial sink | Included in denitrification estimates | [35] |
| Sediment burial | Natural | Relatively minor | [28] |
Phosphorus (P)
Major sources of marine phosphorus
| Source | Type | Approximate flux | Main references |
|---|---|---|---|
| River discharge | Mixed natural + anthropogenic | 2–5 Tg P yr−1 | [10] |
| Estuarine desorption from suspended particles | Natural process enhanced by anthropogenic loading | Often 2–5× dissolved river phosphate input | [44] |
| Atmospheric deposition | Mixed | Relatively small globally | [62] |
| Submarine groundwater discharge | Mixed | Uncertain; regionally important | [63] |
| Sediment recycling under hypoxia | Internal source | Regionally very important | [47] |
Major sinks of marine phosphorus
| Sink | Type | Approximate flux | Main references |
|---|---|---|---|
| Burial in marine sediments | Natural | 3–10 Tg P yr−1 | [48] |
| Authigenic mineral formation | Natural | Included in burial estimates | [44] |
| Phosphine (PH3) production | Possible minor sink | Highly uncertain | [49] |
Silicon (Si)
Major sources of marine silicon
| Source | Type | Approximate flux | Main references |
|---|---|---|---|
| River discharge | Natural dominant source | Major component of 340–490 Tg Si yr−1 total input | [2] |
| Submarine groundwater discharge | Natural | Regionally important | [2] |
| Hydrothermal inputs | Natural | Moderate | [2] |
| Aeolian dust | Natural | Relatively small | [2] |
| Glacial weathering | Natural | Increasing importance in polar regions | [64] |
| Upwelling from deep ocean | Internal recycling | Very large internal flux | [21] |
Major sinks of marine silicon
| Sink | Type | Approximate flux | Main references |
|---|---|---|---|
| Burial of biogenic silica | Natural | ~250 Tg Si yr−1 | [2][65] |
| Reverse weathering | Natural | Major sink | [51] [2] |
| Sponge silica accumulation | Natural | Smaller but significant | [66] |
| Reservoir trapping behind dams | Anthropogenic | Regionally important | [20] [54] |
Related articles
- What causes eutrophication?
- Possible consequences of eutrophication
- Marine microorganisms
- Plankton bloom
- Open ocean habitat
- Deep sea habitat
- Ocean carbon sink
- Blue carbon sequestration
- Ocean acidification
- Other articles in the Category:Eutrophication.
References
- ↑ 1.0 1.1 Beusen, A. H. W., Bouwman, A. F., Van Beek, L. P. H., Mogollón, J. M. and Middelburg, J.J. 2015. Global riverine N and P transport to ocean increased during the twentieth century despite increased retention along the aquatic continuum. Biogeosciences Discuss. 12: 20123–20148
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 Tuholske, C., Halpern, B.S., Blasco, G., Villasenor, J.C., Frazier, M. and Caylor, K. 2021. Mapping global inputs and impacts from human sewage in coastal ecosystems. PLoS ONE 16(11), e0258898 Cite error: Invalid
<ref>tag; name "T21" defined multiple times with different content - ↑ Houlton, B.Z., Morford, S.L. and Dahlgren, R.A. 2018. Convergent evidence for widespread rock nitrogen sources in Earth’s surface environment. Science 360: 58–62
- ↑ Davies‐Barnard, T. and Friedlingstein, P. 2020. The global distribution of biological nitrogen fixation in terrestrial natural ecosystems. Global Biogeochemical Cycles 34, e2019GB006387
- ↑ 5.0 5.1 5.2 Beusen, A.H.W. and Bouwman, A.F. 2022. Future projections of river nutrient export to the global coastal ocean show persisting nitrogen and phosphorus distortion. Front. Water 4, 893585
- ↑ 6.0 6.1 6.2 6.3 Malone, T.C. and Newton, A. 2020. The globalization of cultural eutrophication in the coastal ocean: Causes and consequences. Front. Mar. Sci. 7: 670
- ↑ Yamamoto, A., Hajima, T., Yamazaki, D., Aita, M.N., Ito, A., Kawamiya, M. 2022. Competing and accelerating effects of anthropogenic nutrient inputs on climate-driven changes in ocean carbon and oxygen cycles. Science Advances 8, eabl9207
- ↑ Tivig, M., Keller, D.P. and Oschlies, A. 2021. Riverine nitrogen supply to the global ocean and its limited impact on global marine primary production: a feedback study using an Earth system model. Biogeosciences 18: 5327–5350
- ↑ 9.0 9.1 9.2 Micella, I., Kroeze, C., Bak, M.P. and Strokal, M. 2024. Causes of coastal waters pollution with nutrients, chemicals and plastics worldwide. Marine Pollution Bulletin 198, 115902
- ↑ 10.0 10.1 Seitzinger, S.P., Mayorga, E., Bouwman, A.F., Kroeze, C., Beusen, A.H.W., Billen, G., Van Drecht, G., Dumont, E., Fekete, B.M., Garnier, J. and Harrison, J.A. 2010. Global river nutrient export: A scenario analysis of past and future trends. Global Biogeochemical Cycles 24, GB0A08
- ↑ Wilson, S.J. et al. 2024. Global subterranean estuaries modify groundwater nutrient loading to the ocean. Limnology and Oceanography Letters 9, 411–422
- ↑ North Sea Task Force 1993. North Sea Quality Status Report. Oslo and Paris Commissions.
- ↑ Rendell, A. R., Ottley, C. J., Jickells, T. D. and Harrison, R. M. 1993. Atmospheric nitrogen inputs to the North Sea. Tellus 45B: 53–63
- ↑ Hallstrøm, S., Benavides, M., Salamon, E.R., Arístegui, J. and Riemann, L. 2022. Activity and distribution of diazotrophic communities across the Cape Verde Frontal Zone in the Northeast Atlantic Ocean. Biogeochemistry 160: 49–67
- ↑ Martínez-Pérez, C., Mohr, W., Löscher, C. et al. 2016. The unicellular diazotrophic symbiont UCYN-A is a key player in the marine nitrogen cycle. Nat. Microbiol. 1, 16163
- ↑ 16.0 16.1 Galloway, J. N. et al. 2004. Nitrogen cycles: past, present, and future. Biogeochemistry 70: 153–226
- ↑ 17.0 17.1 17.2 17.3 Voss, M., Bange, H.W., Dippner, J.W., Middelburg, J.J., Montoya, J.P. and Ward, B. 2013. The marine nitrogen cycle: recent discoveries and uncertainties. Phil. Trans. R. Soc. B 368: 20130121
- ↑ Tang, W., Li, Z. and Cassar, N. 2019. Machine learning estimates of global marine nitrogen fixation. J. Geophys. Res. Biogeosci. 124: 717–730
- ↑ Benavides, M., Bonnet, S., Berman-Frank, I. and Riemann, L. 2018. Deep ocean nitrogen fixation. Front. Mar. Sci. 5: 108
- ↑ 20.0 20.1 20.2 Ittekkot, V., Humborg, C. and Schaefer, P. 2000. Hydrological alterations and marine biogeochemistry: a silicate issue? BioScience 50: 776–782
- ↑ 21.0 21.1 21.2 21.3 Sarmiento, J.L. and Gruber, N. 2006. Ocean Biogeochemical Dynamics. Princeton University Press
- ↑ 22.0 22.1 De Borger, E., Braeckman, U. and Soetaert, K. 2021. Rapid organic matter cycling in North Sea sediments. Continental Shelf Research 214: 104327
- ↑ Wollast, R. 1998. Evaluation and comparison of the global carbon cycle in the coastal zone and in the open ocean. In: Brink, K.H., Robinson, A. (Eds.), The Sea. John Wiley & Sons, Inc., Hoboken, New Jersey, pp. 213–252
- ↑ Huettel, M., Berg, P. and Kostka, J.E. 2014. Benthic exchange and biogeochemical cycling in permeable sediments. Ann. Rev. Mar. Sci. 6: 23–51
- ↑ Braeckman, U., Foshtomi, M.Y., Van Gansbeke, D., Meysman, F., Soetaert, K., Vincx, M. and Vanaverbeke, J. 2014. Variable importance of macrofaunal functional biodiversity for biogeochemical cycling in temperate coastal sediments. Ecosystems 17: 720–737
- ↑ Toussaint, E., De Borger, E., Braeckman, U., De Backer, A., Soetaert, K. and Vanaverbeke, J. 2021. Faunal and environmental drivers of carbon and nitrogen cycling along a permeability gradient in shallow North Sea sediments. Science of the Total Environment 767: 144994
- ↑ Canfield, D., Jørgensen, B., Fossing, H., Glud, R., Gundersen, J., Ramsing, N., Thamdrup, B., Hansen, J., Nielsen, L. and Hall, P.O. 1993. Pathways of organic carbon oxidation in three continental margin sediments. Mar. Geol. 113: 27–40
- ↑ 28.0 28.1 Middelburg, J.J., Soetaert, K., Herman, P.M.J. and Heip, C.H.R. 1996. Denitrification in marine sediments: a model study. Global Biogeochem. Cycles 10: 661–673
- ↑ 29.0 29.1 Tappin, A.D. 2000. An examination of the fluxes of nitrogen and phosphorus in temperate and tropical estuaries: current estimates and uncertainties. Estuarine, Coastal and Shelf Science 55: 885–901
- ↑ 30.0 30.1 30.2 30.3 30.4 30.5 Seitzinger, S., Harrison, J.A., Böhlke, J.K., Bouwman, A.F., Lowrance, R., Peterson, B., Tobias, C. and Van Drecht, G. 2006. Denitrification across landscapes and waterscapes: A synthesis. Ecological Applications 16: 2064–2090
- ↑ Malcolm, S.J. and Sivyer, D.B. 1997. Nutrient recycling in intertidal sediments. In: Jickells, T. and Rae, J.E. (Eds.), Biogeochemistry of Intertidal Sediments. Cambridge University Press, pp. 59–83
- ↑ Burgin, A. and Hamilton, S. 2007. Have we overemphasized the role of denitrification in aquatic ecosystems? A review of nitrate removal pathways. Frontiers in Ecology and the Environment 5: 89–96
- ↑ Rysgaard, S. B., Christensen, P.B. and Nielsen, L. P. 1995. Seasonal variation in nitrification and denitrification in estuarine sediment colonized by benthic microalgae and bioturbating fauna. Marine Ecology Progress Series 126: 111–121
- ↑ Burgin, A.J. and Hamilton, S.K. 2007.Have we overemphasized the role of denitrification in aquatic ecosystems? A review of nitrate removal pathways. Frontiers in Ecology and the Environment 5: 89–96
- ↑ 35.0 35.1 Thamdrup, B. and Dalsgaard, T. 2002. Production of N2 through anaerobic ammonium oxidation coupled to nitrate reduction in marine sediments. Applied and Environmental Microbiology 68: 1312–1318
- ↑ 36.0 36.1 Deutsch, C., Gruber, N., Key, R. M. and Sarmiento, J. L. 2001. Denitrification and N2 fixation in the Pacific Ocean. Global Biogeochemical Cycles 15: 483–506
- ↑ Zheng, Y., Hou, L., Zhang, Z., Ge, J., Li, M., Yin, G., Han, P., Dong, H., Liang, X., Gao, J., Gao, D. and Liu, M. 2021. Overlooked contribution of water column to nitrogen removal in estuarine turbidity maximum zone (TMZ). Science of the Total Environment 788: 147736
- ↑ O'Connor, J.A., Erler, D.V., Ferguson, A. and Mahler, D.Y. 2022. The tidal freshwater river zone: Physical properties and biogeochemical contribution to estuarine hypoxia and acidification - The “hydrologic switch”. Estuarine, Coastal and Shelf Science 268, 107786
- ↑ Laverman, A.M., Morelle, J., Roose-Amsaleg, C. and Pannard, A. 2021. Estuarine benthic nitrate reduction rates: Potential role of microalgae? Estuarine, Coastal and Shelf Science 257, 107394
- ↑ Fortune, J., Butler, E.C.V. and Gibb, K. 2023. Estuarine benthic habitats provide an important ecosystem service regulating the nitrogen cycle. Marine Environmental Research 190, 106121
- ↑ Veraart, A.J., de Klein, J.J. and Scheffer, M. 2011. Warming can boost denitrification disproportionately due to altered oxygen dynamics. PLoS One 6(3): e18508
- ↑ Marks, B.M., Chambers, L. and White, J.R. 2016. Effect of fluctuating salinity on potential denitrification in coastal wetland soil and sediments. Soil Science Society of America Journal 80: 516–526
- ↑ Dai, M., Zhao, Y., Cha,i F., Chen, M., Chen, N., Chen, Y., Cheng, D., Gan, J., Guan, D., Hong, Y., Huang, J., Lee, Y., Leung, K.M.Y., Lim, P.E., Lin, S., Lin, X., Liu, X., Liu, Z., Luo, Y-W., Meng, F., Sangmanee, C., Shen, Y., Uthaipan, K., Wan Talaat, W.I.A., Wan, X.S., Wang, C., Wang, D., Wang, G., Wang, S., Wang, Y., Wang, Y., Wang, Z., Wang, Z., Xu, Y., Yang, J-Y.T,, Yang, Y., Yasuhara, M., Yu, D., Yu, J., Yu, L., Zhang, Z. and Zhang, Z. 2023. Persistent eutrophication and hypoxia in the coastal ocean. Cambridge Prisms: Coastal Futures 1, e19, 1–28
- ↑ 44.0 44.1 44.2 44.3 44.4 Paytan, A. and McLaughlin, K. 2007. The oceanic phosphorus cycle. Chem. Rev. 107: 563–576
- ↑ Krom, M.D. and Berner, R.A. 1980. Adsorption of phosphate in anoxic marine sediments. Limnol. Oceanogr. 25: 797–806
- ↑ Frossard, E., Brossard, M., Hedley, M.J. and Metherell, A. 1995. Reactions controlling phosphorus cycling in soils. In: Tiessen, H. (Ed.), Phosphorus in the Global Environment. John Wiley & Sons, pp. 107–138
- ↑ 47.0 47.1 Middelburg, J.J. and Levin, L.A. 2009. Coastal hypoxia and sediment biogeochemistry. Biogeosciences 6: 1273–1293
- ↑ 48.0 48.1 Benitez-Nelson, C.R. 2000. The biogeochemical cycling of phosphorus in marine systems. Earth-Science Reviews 51: 109–135
- ↑ 49.0 49.1 Gassman, G. 1994. Phosphine in the fluvial and marine hydrosphere. Marine Chemistry 45: 197–205
- ↑ Van Cappellen, P., Dixit, S. and van Beusekom, J. 2002. Biogenic silica dissolution in the oceans: reconciling experimental and field-based dissolution rates. Global Biogeochemical Cycles 16, 1075
- ↑ 51.0 51.1 Michalopoulos, P., Aller, R.C. and Reeder, R.J. 2000. Conversion of diatoms to clays during early diagenesis in tropical continental shelf muds. Geology 28: 1095–1098
- ↑ Simpson, T.L. and Volcani, B.E. 1981. Silicon and siliceous structures in biological systems. Springer-Verlag, New York
- ↑ Tréguer, P. J. and De La Rocha, C. L. 2013. The world ocean silica cycle. Annu. Rev. Mar. Sci. 5: 477–501
- ↑ 54.0 54.1 Humborg, C., Conley, D.J., Rahm, L., Wulff, F., Cociasu, A. and Ittekkot, V. 2000. Silicon retention in river basins: far-reaching effects on coastal marine ecosystems. Ambio 29: 45–50
- ↑ Bopp, L., Aumont, O., Cadule, P., Alvain, S. and Gehlen, M. 2005. Response of diatom distribution to global warming. Geophys. Res. Lett. 32
- ↑ Laufkötter, C. et al. 2015. Drivers and uncertainties of future global marine primary production. Biogeosciences 12: 6955–6984
- ↑ 57.0 57.1 57.2 57.3 Morel, F.M.M. and Price, N.M. 2003. The biogeochemical cycles of trace metals in the oceans. Science 300: 944–947
- ↑ Duce, R.A. et al. 2008. Impacts of atmospheric anthropogenic nitrogen on the open ocean. Science 320: 893–897
- ↑ Tang, W., Li, Z. and Cassar, N. 2019. Machine learning estimates of global marine nitrogen fixation. J. Geophys. Res. Biogeosci. 124: 717–730
- ↑ Benavides, M., Bonnet, S., Berman-Frank, I. and Riemann, L. 2018. Deep ocean nitrogen fixation. Front. Mar. Sci. 5: 108
- ↑ Wilson, S.J. et al. 2024. Global subterranean estuaries modify groundwater nutrient loading to the ocean. Limnology and Oceanography Letters 9: 411–422
- ↑ Mahowald, N. et al. 2008. Global distribution of atmospheric phosphorus sources, concentrations and deposition rates, and anthropogenic impacts. Global Biogeochemical Cycles 22: GB4026
- ↑ Slomp, C.P. and Van Cappellen, P. 2004. Nutrient inputs to the coastal ocean through submarine groundwater discharge: controls and potential impact. Journal of Hydrology 295: 64–86
- ↑ Hawkings, J.R. et al. 2017. Ice sheets as a significant source of highly reactive nanoparticulate iron to the oceans. Nature Communications 5: 3929
- ↑ Tréguer, P.J. and De La Rocha, C.L. 2013. The world ocean silica cycle. Annual Review of Marine Science 5: 477–501
- ↑ Maldonado, M. et al. 2019. Sponge skeletons as an important sink of silicon in the global oceans. Nature Geoscience 12: 815–822
Please note that others may also have edited the contents of this article.
|