Common biomarkers for the assessment of marine pollution
One way to assess the health of an ecosystem is to use of biomarkers to assess marine pollution. This article looks at a number of suitable biomarkers that can be used on bivalves or fish to assess the level of marine pollution.
Contents
Introduction
The use of biomarkers stands for a fundamental approach in the assessment of ecosystem health. It allows the early detection of biological changes due to exposure to chemical pollutants, which may result in long-term physiological disturbances. Mussels, such as Mytilus edulis and other marine bivalves, as well as fish (e.g. Mullus sp., Platichthys flesus L., Zoarces viviparus, Perca sp.) are widely used in monitoring programs as sensitive indicators, so-called bioindicators, of the exposure to, and the biological effects of metals and organic pollutants. [1] [2] [3] [4] [5] [6] [7] The choice of bivalve molluscs in monitoring programs is based on their wide geographic distribution, their abundance and accessability in the field as well as in aquaculture. In the case of fish, even though the sampling is expensive, the importance of their use is linked to their position in the trophic chain and their high commercial value. Both bivalves and fish strongly accumulate organic and anorganic pollutants.
Contrary to chemical monitoring of organisms, which principally evaluates the presence of pollutants in tissues by chemical analysis, biomonitoring methods evaluate not only the presence, but what is more significant, the response of the organisms to these pollutants by the assessment of biomarkers, i.e. parameters that reflect their effects at the molecular, cellular, organ, and organism level. The use of these biomarkers in monitoring does not replace chemical monitoring or population studies, but it integrates them in determining the toxic effects of pollutants, also when they are present at low, sub-lethal concentrations.
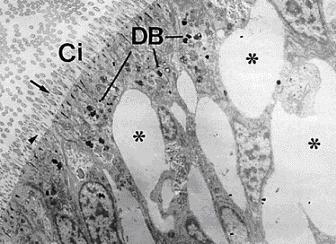
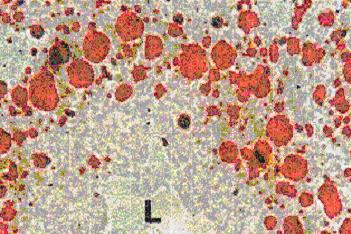
For example, initial studies concerning the toxic effects of pollutants on the cell structure and function showed morphological aberrations (Fig. 1) and changes on cell organelles, such as lipid inclusions (Fig. 2). Thes parameters are now used in monitoring programs. Some biomonitoring strategies involve the assessment of biomarkers in bioindicator organisms. Two main features characterize the suitability of biomarkers for use in large research and monitoring projects (for example MED POL of United Nations, BEEP project of EU, etc.). These are a low cost combined with non-complicated and non-expensive research equipment and most important, the availability of the techniques in routine laboratories.
Suitable biomarker parameters that are measured in bivalves and fish as bioindicator organisms are presented below. There are two main groups: biomarkers that reflect exposure and effect related biomarkers. The latter can be subdivided in biomarkers of physiological stress, of morphological damage, of genotoxicity, and of reproductive toxicity.
Biomarkers of exposure
Biomarkers of exposure include parameters that reflect exposure to a specific class of pollutants, i.e. the biomarkers of this class are specific. Tissue levels of metallothioneins, [8] [9] inhibition of cholinesterase activity, [10] [11] peroxisomal proliferation [12] [13] mixed function oxygenases, [14] [15] [16] [17] and EROD activity [18] are the main biomarkers of this category.
For example, metallothioneins (MTs) are cytosolic proteins with a high affinity for IB and IIB metal ions. They are normally expressed in animal tissues and are involved in heavy metal homeostasis. In an environment with high metal concentrations MTs are over-expressed [8] [18]. This has been observed, among others, in fish and mussels. [18] [19] [20] Therefore, MTs are indicators of metal contamination and are widely used as a tool for coastal biomonitoring programs[21][22][23][24][25], such as the 5 year monitoring program in Thermaic Gulf[26].
Biomarkers of morphological damage
A number of histological biomarkers reflect morphological alterations due to contaminants, [27] [28] [29] [30] such as thinning of the epithelium, increase in the number of epithelial basophil cells, changes in the ratio of basophil to digestive cells, the ratio between the lysosome/cytoplasm volumes.
A reduction of the digestive epithelium thickness was the main difference between exposed and reference groups. A reduction or loss of digestive synchrony was also noted after exposure to pollutants. [27] Changes in the proportion of digestive tubule phases, thinning of the digestive epithelium and increased volume density of basophilic cells was noted in mussels exposed to the water accommodated fraction of different oils. [28] [29] Similarly, an increase in the number of epithelial basophil (secretory) cells was observed and the appearance of lipid vacuoles in both digestive and basophil cells.
Biomarkers of stress
Biomarkers related to physiological stress indicate the effects of a wide range of pollutants; they are not substance specific. Parameters are : response to other stressors, and histological alterations.
Response to chemical and non-chemical stress
This category refers to parameters able to indicate whether an there is an effect at organism level. The biomarkers stress on stress [31] [32] and the scope for growth [33] [34] are the main representative of this class.
The simple biomarker stress on stress provides information about the effects of environmental stressors to the organism. The parameter indicates whether contaminants have affected the capacity of molluscs to survive under stressful condition, such as during exposure to air. Its application is very simple and is calculated by the survival time when animals are exposed to air. [31] [32]
Histological stress parameters
Lysosome membrane stability in cryosections, [35]. [36] lysosome membrane stability-neutral red retention time, [37]. oxidative stress / lipofuscin lysosomal content [38] [8] [39] and neutral lipid accumulation[40][41] are the main biomarkers of this category.
The assessment of histological biomarkers are assessed using histochemical / cytochemical techniques, which offers the advantage of the the possibility to analyse a number of other parameters in parallel, such as lipofuscin and unsaturated neutral lipid content (Figures 1 and 2), as well as changes in basic proteins in situ.
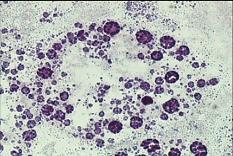
A characteristic example of this class is lysosomal membrane stability in cryosections (LMS). [42] [43] The biomarker is related to changes due to pollution in the membranes of the lysosomes, which constitute main sites of toxic metal and organic pollutant’s sequestration and detoxification. [36][44] This biomarker is mainly applied on digestive gland cryosections of mussels and is based on the cytochemical detection of the lysosomal enzyme N-acetyl-β-hexosaminidase and on the time of acid labilization treatment required to produce lysosomal maximum staining intensity (see figure 3). Minor values of the technique characterize relatively polluted marine areas, while higher ones less polluted areas.
Biomarkers of genotoxicity
DNA damage
Usually includes biomarkers related to the single and double strand breakages, modified bases, DNA-DNA crosslinks, and DNA-protein crosslinks. Alkaline elution and COMET assay are two of the well known biomarkers of this category. [45] [46] [47] [48] [49] [50]
Micronucleus formation
The micronucleus(MN) test is a cytogenetic technique commonly used for the evaluation of genotoxic effects caused by chemical stressors. Micronuclei are formed during cell division when complete or fragment chromosomes fail to be incorporated in the daughter nuclei, forming small additional nuclei. The test consists in the scoring of cells containing one or more cytosolic micronuclei in addition to the main nucleus.[46][51][52] as well as in in different cell types of fishes. [53] [54] [55]
Biomarkers of reproductive impairment.
A variety of compounds may affect reproduction through interference with the hormonal system: the endocrine disruptors. Histological and biochemical biomarkers reflect the effects of these substances: .
Histological parameters for assesment of changes in gonad development are often used in both molluscs and fish studies. Biomarkers assessing gonad (sex gland) development (such as ‘simple gonad index’ or ‘changes in structure of gonad tissue’) are helpful, giving indications of the effects of pollution on the reproductive performance of animals. [56] [57] [58] [59] [60]
Biochemical biomarkers for endocrine disruption are ‘vitellogenin levels’, ‘zona radiata proteins’ (zona radiata=radiately striated membrane situated next to the yolk of an ovum)[61] [62] and ‘steroid hormones balance’ [63] have been regarded as useful tools for an assessment of endocrine disruption caused by chemicals in fish. [64] [65]
Related articles
- Biomonitoring of pollution impacts in the marine environment
- Endocrine disrupting compounds in the coastal environment
- Biomarker
- Bioindicator
- Endocrine disrupting compounds
References
- ↑ Krishnakumar, P.K., Casillas, E., Varanasi, U., 1994. Effect of environmental contamination on the health of Mytilus edulis from Puget Sound, Washington: cytochemical measures of lysosomal responses in the digestive cells using automatic image analysis. Marine Ecology Progress Series 106, 249–261.
- ↑ Regoli, F., Orlando, E., 1994a. Seasonal variation of trace metal concentrations in the digestive gland of the Mediterranean mussel Mytilus galloprovincialis: comparison between a polluted and a non-polluted site. Archives of Environmental Contamination andToxicology 27, 36–43.
- ↑ Regoli, F., Orlando, E., 1994b. Accumulation and subcellular distribution of metals (Cu, Fe, Mn, Pb and Zn) in the Mediterranean mussel Mytilus galloprovincialis during a field transplant experiment. Marin Pollution Bulletin 28 (10), 592–600.
- ↑ Walsh, K., Dunstan, R.H., Murdoch, R.N., 1995. Differential bioaccumulation of heavy metals and organopollutants in the soft tissue and shell of the marine gastropod,Austrocochlea constricta. Archives of Environmental Contamination and Toxicology 28, 1095 35–39.
- ↑ Viarengo, A., Ponzano, E., Dondero, F., Fabbri, R., 1997a. A simple spectrophotometric method for metallothionein evaluation in marine organisms: an application to Mediterranean and Antarctic molluscs. Marine Environmental Research 44 (1), 69–84.
- ↑ Regoli, F., 1998. Trace metals and antioxidant enzymes in gills and digestive gland of the Mediterranean mussel Mytilus galloprovincialis. Archives of Environmental Contamination and Toxicology 991 34, 48–63.
- ↑ Lafontaine, de Y., Gagne, F., Blaise, C., Costan, G., Gagnon, P., Chan, H.M., 2000. Biomarkers in zebra mussels (Dreissena polymorpha) for the assessment and monitoring of water quality of the St Lawrence river (Canada). Aquatic Toxicology 50, 51–71.
- ↑ 8.0 8.1 8.2 Viarengo, A., 1989. Heavy metals in marine invertebrates: mechanisms of regulation and toxicity at the cellular level. CRC Critical Reviews in Aquatic Science 1, 295–317.
- ↑ Viarengo, A., Burlando, B., Dondero, F., Marrò, A., Fabbri, R., 1999b. Metallothionein as a tool in biomonitoring programmes. Biomarkers 4, 455-466.
- ↑ Sturm, A., da Silva de Assis, H.C., Hansen, P.D., 1999. Cholinesterases of marine teleost fish: enzymological characterization and potential use in the monitoring of neurotoxic contamination. Mar. Environ. Res. 47, 389-398.
- ↑ Rodriguez-Fuentes, G., Gold-Bouchot, G., 2004. Characterization of cholinesterase activity from different tissues of Nile tilapia (Oreochromis niloticus). Mar. Environ. Res. 58, 505-509.
- ↑ Cajaraville, M.P., Cancio, I., Ibabe, A., Orbea, A., 2003. Peroxisome proliferation as a biomarker in environmental pollution assessment. Microsc. Res. Tech. 61, 191-202.
- ↑ Orbea, A., Cajaraville, M.P., 2006. Peroxisome proliferation and antioxidant enzymes in transplanted mussels of four basque estuaries with different levels of polycyclic aromatic hydrocarbon and polychlorinated biphenyl pollution. Environ. Toxicol. Chem. 25, 1616-1626.
- ↑ Payne, J.F., 1976. Field evaluation of benzopyrene hydroxylase induction as a monitor for marine petroleum pollution. Science 191, 945-946.
- ↑ Stegeman, J.J., Hahn, M.E., 1994. Biochemistry and molecular biology of monooxygenase: current perspective on forms, functions, and regulation of cytochrome P450 in aquatic species. In: Malins, D.C., Ostrander, G.K., (Eds.), Aquatic toxicology: molecular, biochemical and cellular perspectives. Lewis Publishers, Boca Raton, FL, pp. 87-206.
- ↑ Bucheli, T.D., Fent, K., 1995. Induction of cytocrome P450 as a biomarker for environmental contamination in aquatic ecosystem. Crit. Rev. Environ. Sci. Technol. 25, 201-268.
- ↑ Goeptar, A.R., Scheerens, H., Vermeulen, N.P.E., 1995. Oxygen reductase and substrate reductase activity of cytochrome P450. Crit. Rev. Toxicol. 25, 25-65.
- ↑ 18.0 18.1 18.2 Viarengo, A., Bettella, E., Fabbri, R., Burlando, B., Lafaurie, M., 1997b. Heavy metal inhibition of EROD activity in liver microsomes from the bass Dicentrarchus labrax exposed to organic xenobiotics: role of GSH in the reduction of heavy metal effects. Mar. Environ. Res. 44, 1-11.
- ↑ Bremner, I., 1987. Interactions between metallothionein and trace elements. Prog. Food Nutr. Sci. 11, 1-37.
- ↑ Webb, M., 1987. Toxicological significance of metallothinein. EXS 52, 109-134.
- ↑ Hamilton, S.J., Mehrle, P.M. ,1986, Metallothionein in Fish: Review of Its Importance in Assessing Stress from Metal Contaminants. Transactions of the American Fisheries Society . Vol. 115 (4) 596-609
- ↑ Olsson, P.-E. ,1996, Metallothioneins in fish: induction and use in environmental monitoring . In: Toxicology of Aquatic Pollution. Physiological, Molecular and Cellular Approaches. Society for Experimental Biology Seminar Series. 57: 187-204
- ↑ Hauser-Davis R.A., de Campos R.C., Ziolli R.L.,2012, Fish metalloproteins as biomarkers of environmental contamination. Rev Environ Contam Toxicol. 218:101-23
- ↑ Ramšak A., Ščančar J., Horvat M.,2012, Evaluation of Metallothioneins in Blue Mussels (Mytilus galloprovincialis) as a Biomarker of Mercury and Cadmium. Exposure in the Slovenian waters (Gulf of Trieste): A Long-term Field Study . Acta Adriatica 53(1): 71 - 86
- ↑ Viarengo A, Ponzano E,Dondero F, Fabbrith R.,1997, A Simple Spectrophotometric Method for Metallothionein Evaluation in Marine Organisms: an Application to Mediterranean and Antarctic Molluscs. Marine Environmental Research, Vol. 44, No. 1, pp. 69-84, 1997
- ↑ E.K. Raftopoulou & V.K. Dimitriadis (2010) Assessment of the health status of mussels Mytilus galloprovincialis along Thermaikos Gulf (Northern Greece): An integrative biomarker approach using ecosystem health indices. Ecotoxicology and Environmental Safety. Vol 73 (7):1580–1587
- ↑ 27.0 27.1 Lowe, D.M., Moore, M.N., Clarke, R.K., 1981. Effects of oil on digestive cells in mussels:quantitative alterations in cellular and lysosomal structure. Aquat. Toxicol. 1, 213-226.
- ↑ 28.0 28.1 Lowe, D. M., 1988. Alteration in the cellular structure of Mytilus edulis resulting from exposure to environmental contaminants under field and experimental conditions. Mar. Ecol. Prog.Ser. 46, 91-100.
- ↑ 29.0 29.1 Cajaraville, M.P., Marigomez, J.A., Angulo, E., 1990. Short-term toxic effects of 1-naphthol on the digestive gland-gonad complex of the marine prosobranch Littorina littorea (L): a light microscopic study. Arch. Environ. Contam. Toxicol. 19, 17-24. 37.
- ↑ Cajaraville, M.P., Marigomez, J.A., Diez, G., Angulo, E., 1992. Comparative effects of the water accommodated fraction of three oils on mussels. 2. Quantitative alterations in the structure of the digestive tubules. Comp. Biochem. Physiol. C 102, 113-123.
- ↑ 31.0 31.1 Viarengo, A., Canesi, L., Pertica, M., Mancinelli, G., Accomando, R., Smaal, A.C., Orunesu, M., 1995. Stress on stress response: a simple monitoring tool in the assessment of general stress syndrome in mussels. Mar. Environ. Res. 39, 245–248.
- ↑ 32.0 32.1 Pampanin, D.M., Volpato, E., Marangon, I., Nasci, C., 2005. Physiological measurements from native and transplanted mussel (Mytilus galloprovincialis) in the canals of Venice. Survival in air and condition index. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 140, 41-52.
- ↑ Widdows, J., Donkin, P., 1992. Mussels and environmental contaminants: bioaccumulation and physiological aspects. In: Gosling, E., (Ed.), The mussel Mytilus: ecology,physiology, genetics and culture. Elsevier, Amsterdam, pp. 383-424.
- ↑ Widdows, J., Donkin, P., Staff, F. J., Matthiessen, P., Law, R. J., Allen, Y. T., Thain, J. E., Allchin, C. R., Jones, B. R., 2002. Measurement of stress effects (scope for growth) and contaminant levels in mussels (Mytilus edulis) collected from the Irish Sea. Mar. Environ. Res. 53, 327-356.
- ↑ Moore, M.N., 1985. Cellular responses to pollutants. Marine Pollution Bulletin 16, 134–139
- ↑ 36.0 36.1 Viarengo, A., Moore, M.N., Mancinelli, G., Mazzucotelli, A., Pipe, R.K., Farrar, S.V., 1987. Metallothioneins and lysosomes in metal toxicity and accumulation in marine mussels: the effect of cadmium in the presence and absence of phenanthrene. Marine Biology 94, 251–257.
- ↑ Lowe, D.M., Pipe, R.K., 1994. Contaminant induced lysosomal membrane damage in marine mussel digestive cells: an in vitro study. Aquat. Toxicol. 30, 357-365
- ↑ George, S.G., Viarengo, A., 1985. A model for heavy metal homeostasis and detoxication in mussel. Mar. Pollut. Physiol.: Recent advances, 125-143.
- ↑ Viarengo, A., Nott, J.A., 1993. Mechanisms of heavy metal cation homeostasis in marine invertebrates. Comparative Biochemistry and Physiology 104, 355–372.
- ↑ Koehler, A., Deisemann, H., Lauritzen, B., 1992. Histological and cytochemical indices of toxic injury in the liver of the dab Limanda limanda. Mar. Ecol. Prog. Ser. 91, 141-153.
- ↑ Koehler, A., 2004. Toxic injury and gender-specific hepatocellular carcinogenesis in flounder (Platichthys flesus (L.). Aquat. Toxicol. 70, 257-276.
- ↑ Moore, M.N., 1976. Cytochemical demonstration of latency of lysosomal hydrolases in digestive cells of the common mussel Mytilus edulis, and changes induced by thermal stress. Cell Tissue Research 175 (3), 279–287.
- ↑ UNEP/RAMOGE, 1999. Manual on the biomarkers recommended for the MED POL biomonitoring programme. UNEP, Athens, Greece.
- ↑ Moore, M.N., 1985. Cellular responses to pollutants. Marine Pollution Bulletin 16, 134–139.
- ↑ Batel, R., Vukmirovic, M., Jacsic, Z., Bihari, N., 1994. Impact of pollution on DNA fragmentation in marine invertebrates. Use of Aquatic Invertebrates as Tools for Monitoring the Environmental Hazards, 109-117.
- ↑ 46.0 46.1 Bolognesi, C., Landini, E., Roggieri, P., Fabbri, R., Viarengo, A., 1999. Genotoxicity biomarkers in the assessment of heavy metal effects in mussels: experimental studies. Environ. Mol. Mutagen. 33, 287-292.
- ↑ Bolognesi, C., Frenzilli, G., Lasagna, C., Perrone, E., Roggieri, P., 2004. Genotoxicity biomarkers in Mytilus galloprovincialis: wild versus caged mussels. Mutat. Res. 552, 187-196.
- ↑ Bolognesi, C., Perrone, E., Roggieri, P., Pampanin, D., Sciutto, A., 2006. Assessment of micronuclei induction in peripheral erythrocytes of fish exposed to xenobiotics under controlled conditions. Aquat. Toxicol. 78, S93-98.
- ↑ Lee, R.F., Steinert, S., 2003. Use of the single cell gel electrophoresis/comet assay for detecting DNA damage in aquatic (marine and freshwater) animals. Mutat. Res. 544, 43-64.
- ↑ Frenzilli, G., Scarcelli, V., Del Barga, I., Nigro, M., Forlin, L., Bolognesi, C., Sture, J., 2004. DNA damage in Eelpout (Zoarces viviparous) from Goteborg harbour. Mutat. Res. 552, 153-162.
- ↑ Majone, F., Brunetti, R., Fumagalli, O., Gabriele, M., Levis, A.G., 1990. Induction of micronuclei by mitomycin C and colchicine in the marine mussel Mytilus galloprovincialis. Mutat. Res. 244, 147-151.
- ↑ Scarpato, R., Migliore, L., Alfinito-Cognetti, G., Barale, R., 1990. Induction of micronuclei in gill tissue of Mytilus galloprovincialis exposed to polluted marine waters. Mar. Pollut. Bull. 21, 74-80.
- ↑ Al-Sabti, K., Metcalfe, C.D., 1995. Fish micronuclei for assessing genotoxicity in water. Mutat. Res. 343, 121-135.
- ↑ Arkhipchuk, V.V., Garanko, N.N., 2005. Using the nucleolar biomarker and the micronucleus test on in vivo fish fin cells. Ecotoxicol. Environ. Saf. 62, 42-52.
- ↑ Barsiene, J., Lehtonen, K. K., Koehler, A., Broeg, K., Vuorinen, P.J., Lang, T., Pempkowiak, J., Šyvokien_, J., Dedonyte, V., Rybakovas, A., Repeka, R., Vuontisjärvi, H., Kopecka,J., 2006. Biomarker responses in flounder (Platichthys flesus) and mussel (Mytilus edulis) in the Klaipeda-B_ting_ area (Baltic Sea). Mar. Poll. Bull. 53, 422-436.
- ↑ Lowe, D., Pipe, R.K., 1985. Cellular responses in the mussel Mytilus edulis following exposure to diesel oil emulsions: reproductive and nutrient storage cells. Mar. Environ. Res. 17, 234-237.
- ↑ Kime, D.E., 1995. The effects of pollution on reproduction in fish. Rev. Fish Biol. Fish. 5, 52-96.
- ↑ Minier, C., Levy, F., Rabel, D., Bocquené, G., Godefroy, D., Burgeot, T., Leboulenger, F., 2000. Flounder health status in the Seine Bay. A multibiomarker study. Mar. Environ. Res. 50, 373-377.
- ↑ Van der Oost, R., Beyer, J., Vermeulen, N.P.E., 2003. Fish bioaccumulation and biomarkers in environmental risk assessment: a review. Environ. Toxicol. Pharmacol. 13, 57-149.
- ↑ Aarab, N., Lemaire-Gony, S., Unruh, E., Hanson, P.D., Larsen, B.K., Andersen, O.K., Narbonne, J.F., 2006. Preliminary study of responses in mussels (Mytilus edulis)
- ↑ Haux, C., Björnsson, B.T., Förlin, L., Larsson, Å., Deftos, L.J., 1988. Influence of cadmium exposure on plasma calcium, vitellogenin and calcitonin in vitelogenic rainbow trout. Mar.Environ. Res. 24, 199-210.
- ↑ Spies, R.B., Stegeman, J.J., Rice, D.W., Jr., Woodlin, B., Thomas, P., Hose, J.E., Cross, J.N., Prieto, M., 1990. Sublethal responses of Platichtus stellatus to organic contamination in San Francisco Bay with emphasis on reproduction. In: McCarthy, J. F., Shugart, L. R.(Eds.), Biomarkers of Environmental Contamination. CRC Press, Boca Raton, FL, pp. 87-122.
- ↑ Karels, A., Soimasuo, M., Lappivaara, J., Leppänen, H., Aaltonen, T., Mellanen, P., Oikari, A.,1998. Effects of bleached kraft mill effluent on reproductive steroids and liver MFO activity in populations of perch and roach. Ecotoxicology 7, 123-132.
- ↑ Armstrong, D.T., 1990. Environ. stress and ovarian function. Biol. Reprod.34, 29-39.
- ↑ Martin-Skilton, R., Lavado, R., Thibaut, R., Minier, C., Porte, C., 2006. Evidence of endocrine alteration in the red mullet, Mullus barbatus from the NW Mediterranean. Environ. Pollut.141, 60-8.
Please note that others may also have edited the contents of this article.
|