Seagrass meadows
This article describes the habitat of the seagrass meadows. It gives an introduction to the characteristics, distribution, zonation, succession, biota, threats, functioning and adaptations of the organisms that live in seagrass meadows.
Contents
Introduction
Seagrass communities are highly productive and dynamic ecosystems. Seagrasses are not true grasses but rooted vascular (flowering) plants of terrestrial origin that have successfully returned to the sea. This return needs several adaptations that allow them to live in submerged ocean regions. The sediments where they settle on can be muddy, rocky or sandy. Seagrass ecosystems are species-rich and include endangered species such as dugongs and seahorses. They are important for the geomorphology and ecology of coastal ecosystems through processes such as stabilizing sediments, recycling nutrients and providing the base of the oceanic detrital food webs. Although seagrasses cover only about 0.1% of the oceans[1], they represent more than 1 percent of the total marine primary production, acting as a sink for CO2 (see Blue carbon sequestration). Currently, they are facing many threats, due to human activities and natural causes [2].
Distribution
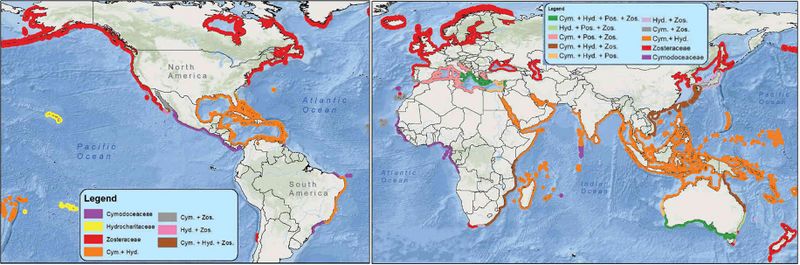
Seagrasses generally inhabit the protected shallow waters of temperate and tropical coastal areas. Seagrass can be patchy, but more often it forms large swaths of vegetation, sometimes over 10,000 km2 in size[3]. The most extensive areas are found in the tropics. The total area covered by seagrass meadows globally is estimated to be on the order of 160,000 km2, but possibly about 100,000 km2 more[4].
Seagrasses belong to 4 families (Cymodoceaceae, Hydrocharitaceae, Posidoniaceae and Zosteraceae). About 70 seagrass species have been reported, which are grouped into 12 genera; three of the genera (Halophila, Zostera and Posidonia), include most of the species[1][5]. A few species occur in colder regions. Four closely related species are native of European waters. There are several distinct areas of seagrass meadows. These areas are the Indo-Pacific region, the seas around Japan and Australia, the central Western Atlantic region, the north East Atlantic region and the Baltic and Mediterranean seas.
- Zostera marina (Zosteraceae) is found from arctic waters along the northern Norwegian coast to the Mediterranean Sea. It is very abundant in the Baltic Sea, the North Sea and along the Atlantic coast to northern Spain.
- Zostera noltii (Zosteraceae) ranges from the southern coast of Norway to the Mediterranean Sea, the Black Sea, the Canary Islands and has even been recorded on the southern coast of Mauretania.
- Cymodocea nodosa (Cymodoceaceae) is found in the Mediterranean Sea, the Canary Islands and the North African coast. It is a warm water seagrass.
- Posidonia oceanica (Posidoniaceae) is only found in the Mediterranean Sea. The distribution is restricted by the mixing zone of Mediterranean water and Atlantic water. Other species may successfully invade in European waters if seeds or fragments are accidentally introduced. Currently, the invasive seagrass
- Halophila stipulacea (Hydrocharitaceae), native to the tropical and subtropical waters of the Red Sea, Persian Gulf and Indian Ocean, is settling in the eastern Mediterranean and has reached the southern coast of Italy. [6]
- Thalassia (Hydrocharitaceae) is the dominant primary producer in tropical coastal seagrass communities, although other macrophytes (seagrasses and macroalgae), benthic and epiphytic diatoms and phytoplankton also contribute to the total community production. The species T. testudinum is found in the Western Atlantic and T. hemprichii in the Indo-Pacific.
Thalassia-dominated meadows are considered to be amongst the most highly productive marine systems on Earth[7].
Multibeam echo sounding (MBES) is currently the most widely used technique for mapping seagrass meadows. Machine learning approaches for data processing are increasingly used for the automatic classification of seagrass species; however, ground-truth data remain essential for validation and accuracy assessment. Multispectral MBES is a promising development that can further facilitate and improve the discrimination of seagrass species.[8]
Requirements for development and restoration
Seagrasses develop under the following conditions[1]:
- Salt or brackish water. Most seagrass species grow well at a salinity level of 10–30 PSU for temperatures around 20oC. [9] Fastest growth rates are recorded for salinities around 20 PSU[10].
- Seagrass needs light for photosynthesis. It is highly sensitive to light conditions and therefore grows in shallow (less than 20 meters) regions along the coasts. The average depth is a few meters, but seagrass has been recorded down to 90 meters depth in clear water. The required amount of light depends on species and temperature. Experiments of Collier et al. (2016[11]) with several tropical species (Cymodocea serrulata, Halodule uninervis, Halophila ovalis and Zostera muelleri) showed that light thresholds that maintained 50% and 80% of seagrass shoot density fell within the ranges 1.1–5.7 and 3.8–10.4, respectively (surface irradiance measured in photosynthetic photon flux density, mol.m-2.day-1). The impact of light limitation on shoot densities and growth rates was higher at warm than at cool temperatures.
- Clear water. The plants die when the water becomes turbid by sediment suspension. Seagrasses globally have light requirements in the range of 4 to 36% of sub-surface irradiance (Zostera muelleri requires 16 to 36% of sub-surface irradiance for survival and Posidonia sinuosa 8 to 12%). If light availability is sustained below this level, complete loss of seagrass is predicted[12]. This contrasts with the general light requirements of phytoplankton which are estimated at about 1%.
- A soft substrate such as mud or sand, but some species can also grow on rocky sediments and corals.
- A gently sloping coast, with little or no tidal currents or strong waves, is preferred.
The environmental tolerance thresholds of seagrass species in different climate zones vary by species. Habitat Suitability Indices that associate seagrass species with suitable habitat characteristics can be helpful to guide seagrass restoration projects[13]. Successful restoration projects show that, in addition to the above requirements, large and densely planted plots are more likely to succeed by overcoming seed recruitment limitation and by stabilizing sediments[14]. The low overall survival rate of many seagrass restoration trials can be explained by their small scale[15]. Seagrass recovery requires a large supply of seeds over an extended period of time, as illustrated by the successful seagrass recovery in Virginia Bay (US East Coast) after ten years of sustained large-scale sowing. Cultivation of seagrass species with large seed production potential in nursery systems, for sowing, planting and growing, is an intermediate step necessary to collect the required amount of seeds[16].
Zonation
Different species grow in different places. Zonation usually follows the ‘classical’ pattern of small and narrow leaved species in the intertidal zone, which in the shallow subtidal zone become replaced by the more broadleaved Thalassia[17].
In areas with consistent disturbance and unstable sediments, which are low in organic content, Syringodium filiforme may be the most abundant seagrass, where it is commonly found in a fringe at beaches[17].
Colonizing species of the high and mid intertidal zone are Halodule wrightii or Halophila decipiens (W. Atlantic) and Cymodocea rotundata, Halodule pinifolia and various Halophila spp (Indo-Pacific). They can be found just below the mangrove vegetation. Sometimes they are accompanied by sand-dwelling seaweeds such as Chondria, Hypnea and Caulerpa.
The mid and low intertidal zones are dominated by the climax vegetation Thalassia on stable substrates. Thalassia cannot tolerate prolonged exposure to high temperatures, nor long-term desiccation on intertidal flats[18]. Thalassia-dominated seagrass meadows are generally multi-taxon conglomerates composed of various species of seagrasses, rooted calcareous and fleshy algae (rhizophytic algae), drifting fleshy or filamentous algae, and epiphytes[17]. These smaller seagrass species or macroalgae (for example sand-dwelling Halimeda spp.) may replace Thalassia in deeper water.
The subtidal fringe is an area with Syringodium isoetifolium (Indo-Pacific). This seagrass has tough, cylindrical leaves. Enhalus acoroides can also be present. This seagrass also has very large and tough leaves. In the subtidal zone, the species Halophila stipulacea and Halodule uninervis occur when the substrate is not yet stabilized, whereas Thalassodendron ciliatum and Enhalus acoroides grow on stabilized substrates (Indo-Pacific). Other seagrasses below the depth range of T. hemprichii are Cymodocea serrulata, and at places Enhalus acoroides. Fine carpets of Halophila spp. (principally H. decipiens, H. engelmannii in the W-Atlantic, and H. decipiens, H. ovalis and H. spinulosa in the Indo-Pacific) can be found extending to 30 – 40 m depth. Deeper down the slopes of the W. Atlantic coasts, Thalassia testudinum is replaced by Syringodium filiforme or Halodule wrightii that tolerate lower light levels[17].
 Thalassia hemprichii. Photo credit Steven Victor. |
 Enhalus acoroides. Photo credit Steven Victor. |
 Cymodocea rotundata. Photo credit Steven Victor. |
 Halophila ovalis. Photo credit Fernando Boisset. |
 Thalassia testudinum. Photo credit Angel Fernandez. |
 Halodule wrightii. Photo credit Diane Littler. |
 Halophila engelmannii. Photo credit Sara Lardizabal. |
 Syringodium filiforme. Photo credit Diane Littler. |
European genera are Zostera, Posidonia and Cymodocea.
Zostera or eelgrass is a small genus of widely distributed seagrass, with Z. marina and Z. noltii occurring in western Europe. It is found on sandy substrates and in estuaries where it is submerged or partially floating. The meadows are important for sediment deposition, substrate stabilization, as substrate for epiphytic algae and micro-invertebrates and as nursery grounds. Z. marina is a subtidal species and may grow down to 10 -15 meters depth. Z. noltii forms dense beds in the muddy sand of intertidal areas. Z. marina has a lower tolerance to desiccation and therefore less adapted to intertidal areas. Z. noltii can also occur in subtidal areas, but is usually outcompeted by other seagrasses.
Posidonia oceanica is endemic to the Mediterranean Sea. It occurs in dense meadows or in bands along channels. Balls of fibrous material, known as egagropili, from the foliage of the plant can be found on adjacent beaches. P. oceanica grows from shallow subtidal waters to depth of 50 - 60 meters in areas with very clear water (see also Mediterranean seagrass ecosystem and Posidonia oceanica (Linnaeus) Delile).
Cymodocea nodosa is another seagrass that occurs in the Mediterranean Sea. It grows on sandy sediments in waters down to 20 meters deep. [6]
 Zostera marina Photo credit Ignacio Barbara. |
 Zostera noltii Photo credit Ignacio Barbara. |
 Posidonia oceanica Photo credit Ignacio Barbara. |
 Posidonia oceanica egagropili Photo credit Isabel Rubio. |
 Cymodocea nodosa Photo credit Ignacio Barbara. |
Succession


The sequence of succession in the seagrass beds generally involves early species that stabilize the sediments and increase the sediment nutrient content, and by doing so allow establishment of later species. In the final state of succession, populations of plants or animals remain stable and exist in balance with each other and their environment. This so-called climax community remains relatively unchanged until destroyed by strong disturbance (e.g. invasive species introduction) or human interference (fishing, dredging, etc.).
The preparatory stage of colonization starts with unicellular algae. These algae are often diatoms that stick the sediment grains together by mucus.
In the tropical Western Atlantic the following step in the succession sequence is the colonization by rhizophytic green algae, mainly species of Caulerpa, Halimeda, Penicillus, Rhipocephalus and Udotea, that use a net of rhizoids to anchor in unconsolidated sediments. These algae supply some amounts of organic matter and nutrients to the sediment, but have limited sediment binding capability.
After a while, colonizing seagrass species start to germinate and grow. These species are especially opportunists that grow fast and have long internodes, providing a fast formation of a network of stolons. They survive well in unstable or depositional environments, thereby further stabilizing the sediment surface. Species are, Halodule wrightii (western Atlantic) and Halophila stipulacea, Halophila ovalis and Halodule uninervis (Indo-Pacific). In the intertidal zone, pools are formed by grazing activities of turtles. The margins of these pools are overgrown by seagrass species. Cymodocea species arrive afterwards and take over from the first colonizers. They have shorter internodes, more fleshy roots and can better fixate the sediments. In some sequences of succession, Syringodium filiforme appears instead of H. wrightii, or S. filiforme colonizes after the latter species, in which case the two species grow intermixed. S. filiforme is the least constant member in the sequence of succession and is frequently absent.
With time and increasing development of the community, Thalassia testudinum colonizes the Caribbean Sea and the Gulf of Mexico, and as far north as Cape Canaveral in Florida region. Its dense leaf canopy and rhizome and root system efficiently trap and retain particles, increasing the organic matter of the sediment and fueling the sedimentary microbial cycles. In the Indo-Pacific, Thalassia hemprichii and Enhalus acoroides are the last to colonize. They have very short internodes, densely branched stolons and numerous fleshy roots. This results in very dense vegetation.
Although the structure of T. testudinum and T. hemprichii communities may vary considerably, mainly because T. hemprichii meadows contain more seagrass species, general patterns in structural changes and processes during succession are similar. With the progression toward a climax community, there is an increase in the belowground biomass of the community as well as the leaf portion exposed in the water column, and more nutrients are sequestered by the seagrasses. The increase in leaf area provides an increase in surface area for colonization by epiphytic algae and fauna, with the surface area of the climax community being many times that of either the pioneer seagrasses or the initial algal colonizers. In addition to providing a substrate, the larger leaf area also increases sediment-trapping effects. The climax species T. testudinum and T. hemprichii (with also E. acoroides in the Indo-Pacific), have the highest leaf area, the highest total biomass, and by far the greatest amount of material in the sediments of any species dominant in the earlier stages of succession[17].
Functioning and adaptation
Seagrasses are basically land plants that returned to sea and secondarily colonized marine habitats. The seagrasses have adapted to the marine environment in several ways:
- Salinity. They are halophytes, with different adaptations to seawater. They can regulate osmotic pressure by sequestering salt ions within the cell wall or vacuoles. When a cell swells due to external osmotic pressure, membrane channels open and allow efflux of osmolytes (carbohydrates and amino acids) which carry water with them, restoring normal cell volume[20].
- Submergence. They are hydrophytes able to grow under submerged conditions. For this reason, the tissues of the seagrasses consist of aerenchyma, spongy tissue that creates spaces or air channels in the leaves. Through this aerenchyma, oxygen can be provided to the submerged parts of the plants. Another adaptation of seagrasses to these conditions is an epidermis with chloroplasts for photosynthesis. This is exceptional because the epidermal cells of other higher plants lack chloroplasts. Above this epidermis, a very thin cuticle is present, enabling the uptake of nutrients through the whole leaf surface. Stomata’s are absent and the vascular bundles are strongly reduced.
- Desiccation. Sulfation facilitates water and ion retention in the cell wall to cope with desiccation and osmotic stress at low tide. Sulfated polysaccharides are also important both in terms of resistance to mechanical stresses and as protection from predators[21].
- Erosion. Seagrasses are resistant to erosion by waves and tidal currents, due to well-developed rhizomes and numerous, fleshy roots that anchor into the substrate. The supple leaves are better resistant to water movement than stiff leaves.
- Pollination. Pollen is released from the flowers in gelatinous clumps that are carried by water currents to the pistils (female reproductive organ).
The major stems of seagrasses are called rhizomes. They grow horizontally, mostly just below the surface of the substrate. Together with the roots, they help stabilizing the substrate. The leaves and the roots function as sediment traps. The roots are thicker and more fleshy than the fibrous roots of terrestrial grasses. They thus provide protection against coastal erosion. Nutrient uptake from the seawater is very efficient. This is called nutrient-stripping. As filtering system, seagrasses fulfil an important role in the quality of the coastal waters. They absorb CO2 and are important primary producers, together with epiphytic algae. They have a shelter function for many organisms and they provide important nursery grounds for commercial fish species. [22]
Seagrasses are also ‘ecosystem engineers’ that are able to adapt their environment: they clarify water by damping wave action and retaining fine sediments and they modify substrate chemistry by oxygenating their rhizosphere[23]. Inversely, human interventions that increase turbidity may provoke interactive and/or cascade effects related to light reduction, oxygen demand, changes in bed sediment chemistry and smothering. These multiple stresses eventually lead to the decline of seagrass meadows.
Biota
In the dense seagrass meadows, a wide variety of organisms are fouling such as hydroids, sponges, bryozoa and seaweeds. These organisms mainly attach on older leaves. Many invertebrate species are dependent on seagrasses and would become extinct or greatly reduced in abundance if the seagrasses disappear. An example of the importance of the seagrasses for the survival and development is the Atlantic bay scallop Argopecten irradians. This scallop needs the seagrass habitat for the development of its larval stage and for the protection of juveniles from predators. The dense meadows can discourage predators from entering it.
Seagrass canopies provide food and shelter against predators for a range of epifaunal organisms, such as amphipods, polychaetes, and gastropods. Among seagrass-associated epifauna, amphipods are one of the most abundant and play a major role in the transfer of energy towards higher trophic levels[24]. Filter feeders are abundant in the sediment. Some examples are scallops, jackknife, clams and sea cucumbers[25].
Most seagrasses are not directly consumed by herbivores, because they are too tough. Species that do graze on the seagrasses are sea urchins, sea turtles, dugongs, manatees, some fishes and waterfowl[1]. Seagrass meadows are an essential habitat for 80% of all dugongs and manatees, for nearly 60% of all sea turtles, for about 13% of all dolphins and porpoises and for about 9% of all sharks and rays[26].
Ecosystem services
Seagrass meadows provide many ecosystem services[1][26]; some major services are:
- Fisheries. Seagrasses support global fisheries by procuring nursery habitats for fish, bivalve and crustacean species. They provide refuge from predation (especially for fish larvae and juvenile invertebrates) and a basal food source in the form of edible blades, detritus, and epiphytic algae [27].
- Climate regulation. Seagrass meadows store large amounts of carbon in the biomass and sediment below[28], see Blue carbon sequestration.
- Biodiversity. Seagrass meadows host a rich marine biodiversity, including protected and charismatic species such as dugongs, sea turtles, sharks and seahorses.
- Ocean acidification buffer. Seagrass meadows regulate the chemical composition of seawater by releasing oxygen and removing carbon dioxide during daylight, oxygenating water and buffering ocean acidification.
- Water filtration. Seagrasses are natural filters that trap sediments, excessive nutrients and chemical contaminants out of the water[29]. The soils beneath seagrass meadows have the potential to act as significant sinks for metal pollution, which, once trapped, are effectively isolated from most components of the ecosystem and likely unavailable to re-enter biological processes if the seagrass beds remain intact and healthy. For example, field observations showed that a large proportion of the metals Pb, Zn and Cd released from a smelter in South Australia into adjacent coastal waters were trapped and sequestered in the extensive littoral seagrass meadows[30]. However, some contaminants, such as Cu, can be toxic to seagrass at high concentrations (> 0.5 mg/l)[31].
- Coastal protection. Seagrass meadows stabilize the substrate, enhance sedimentation and dampen wave activity, thereby helping to mitigate coastal erosion and protect against flooding and storm surges. Field observations in a back-barrier basin on the Atlantic US coast showed that in summer the seagrass meadows reduce waves by 20%, currents by 60% and suspended sediment concentrations by 85%, compared to a neighbouring bare site. In winter, however, the less dense meadow hardly had any significant dampening effect[32]. The articles Nature-based shore protection and Wave damping by vegetation discuss more in detail the coastal protection function of seagrass meadows.
- Disease control. Seagrasses can remove microbiological contamination from the water, thus reducing exposure to bacterial pathogens for fish, humans and invertebrates. They produce secondary metabolites with antibacterial and antifungal activity[33].
- Tourism. Seagrass meadows provide cultural services such as sense of identity for local communities and opportunities for recreational activities (e.g. birdwatching, diving, fishing).
The human benefits of seagrasses are extensive and some may not even be discovered yet. However, not all seagrasses provide the full range of ecosystem services year-round, in every habitat they colonize and in all geographic regions. Differences between species, genera or geographical location must be taken into account[34].
Threats
Decline of the population of seagrasses is a worldwide trend. Almost 20 per cent of global seagrass area has been lost since the late nineteenth century and at least 22 of the world’s 72 seagrass species are in decline[26][35]. Only a small fraction (less than 2%) has been recovered since 1900. Despite overall declines in seagrass meadow area across bioregions, changes in individual meadow area are highly variable. Many meadows in the Temperate North Atlantic East, Mediterranean and Tropical Atlantic bioregions shifted towards positive trends after 1990, explaining the recent increasing trends in those bioregions. However, overall meadow area over the past two decades has remained below 90% of maximum for most meadows[35].
Declining trends are mainly caused by human disturbances:
- Agricultural, urban and industrial runoff causing excessive nutrient and sediment inputs.
- Beam trawl fishing and overfishing
- Aquaculture: shading, eutrophication and sediment deterioration
- Dredging for harbors, ports and shipping lanes
- Harvesting, clearance for beaches
- Shipping: anchoring, pollution
- Introduction of exotic species; accidentally, ballast water, aquaculture, hull fouling
- Tourist facilities
Global warming exposes seagrass meadows more frequently to excessive temperatures (4-5°C above the average of normal summer months). Under such conditions severe impacts or even complete dieback has been observed[36]. Statistical analysis of seagrass monitoring data along the US Atlantic coast showed that summer water temperature one year prior to monitoring was a significant predictor of eelgrass presence (not abundance) on a regional scale. Above average summer temperatures corresponded to a decrease in probability of eelgrass presence (and increased probability of eelgrass absence) the following year[37].
Severe storms can also inflict great damage on seagrass meadows[38].
Multiple stressors generally co-occur spatially and temporally. Aquaculture development, destructive fishing, the introduction of non-native species, and boating are all more likely to take place adjacent to or near port development and urban centres[35].
Eutrophication causing overproliferation of macroalgae harms seagrass directly by competing for resources and space or indirectly by reducing oxygen supply and available light for photosynthesis[39][40].
Human activities that alter fine sediment dynamics are a major cause of seagrass decline. Perturbation of the fine sediment dynamics can affect seagrass growth in several ways: reduced light penetration due to increased turbidity[41], change in the physical and chemical properties of the substrate[42] and the settling of suspended particles onto seagrass that impedes gas exchange with the surrounding water[43]. There is strong evidence for a self-sustaining feedback of relatively dense seagrass via sediment stabilization and accordingly a runaway feedback once the seagrass cover becomes too sparse[41][44]. Thus implies that once a seagrass meadow is lost by disturbance of the fine sediment dynamics, it cannot easily be recovered.
Related articles
- Seagrass recovery and restoration in the Wadden Sea
- Mediterranean seagrass ecosystem
- Posidonia oceanica (Linnaeus) Delile
- Blue carbon sequestration
- Nature-based shore protection
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Spalding, M., Taylor, M., Ravilious, C., Short, F. and Green, E.. 2003. The distribution and status of seagrasses. In: Green, E.P., Short, F. (Eds.), World Atlas of Seagrasses. University California Press, pp. 5–26
- ↑ Denny M.W. Gaines S.D. 2007. Encyclopedia of tidepools & rocky shores. University of California Press. p. 705
- ↑ Hemminga, M.A. and Duarte, C.M. 2000. Seagrass Ecology. Cambridge University Press.
- ↑ McKenzie, L.J., Nordlund, L.M., Jones, B.L., Cullen-Unsworth, L.C., Roelfsema, C. and Unsworth, R.K.F 2020. The global distribution of seagrass meadows. Environ. Res. Lett. 15, 074041
- ↑ [www.worldfloraonline.org World Flora Online]
- ↑ 6.0 6.1 Borum J. et al. 2002. European seagrasses: an introduction to monitoring and management. The M&MS project. p.95
- ↑ Westlake, D.F. 1963. Comparisons of plant productivity. Biol. Rev. 38: 385–425
- ↑ Gumusay, M.U., Bakirman,T., Kizilkaya, I.T. and Aykut, N.O. 2019. A review of seagrass detection, mapping and monitoring applications using acoustic systems. European Journal of Remote Sensing 52: 1-29
- ↑ Nejrup, L.B. and Pedersen, M.F. 2008. Effects of salinity and water temperature on the ecological performance of Zostera marina. Aquat. Bot. 88: 239–246
- ↑ Zhang, Y-H., Yu, B., Liu, Y-C., Ma, W., Li, W-T. and Zhang, P.D. 2022. The influence of decreased salinity levels on the survival, growth and physiology of eelgrass Zostera marina. Marine Environmental Research 182, 105787
- ↑ Collier C.J., Adams, M.P., Langlois, L., Waycott, M., O’Brien, K.R., Maxwell P.S. and McKenzie, L. 2016. Thresholds for morphological response to light reduction for four tropical seagrass species. Ecological Indicators 67: 358–366
- ↑ Ralph, P.J., Durako, M.J., Enriquez, S., Collier, C.J. and Doblin, M.A. 2007. Impact of light limitation on seagrasses. Journal of Experimental Marine Biology and Ecology 350: 176–193
- ↑ Erftemeijer, P.L.A., van Gils, J., Fernandes, M.B., Daly, R., van der Heijden, L. and Herman, P.M.J. 2023. Habitat suitability modelling to improve understanding of seagrass loss and recovery and to guide decisions in relation to coastal discharge. Marine Pollution Bulletin 186 (2023) 114370
- ↑ Orth, R.J., Lefcheck, J.S., McGlathery, K.S., Aoki, L., Luckenbach, M.W., Moore, K.A., Oreska, M.P., Snyder, R., Wilcox, D.J. and Lusk, B. 2020. Restoration of seagrass habitat leads to rapid recovery of coastal ecosystem services. Science Advances 6 : eabc6434
- ↑ van Katwijk, M. M., Thorhaug, A., Marba, N., Orth, R. J., Duarte, C. M., Kendrick, G. A., Althuizen, I. H. J., Balestri, E., Bernard, G., Cambridge, M. L., Cunha, A., Durance, C., Giesen, W., Han, Q., Hosokawa, S., Kiswara, W., Komatsu, T., Lardicci, C., Lee, K. S., Meinesz, A., Nakaoka, M., O’Brien, K., Paling, E. I., Pickerell, C., Ransijn, A. M. A. and Verduin, J. J. 2016. Global review of seagrass restoration and the importance of large-scale planting. J. Appl. Ecol. 53: 567–578
- ↑ Van Katwijk, M.M., Van Tussenbroek, B.I., Hanssen, S.V., Hendriks, A.J. and Hanssen L. 2021. Rewilding the sea with domesticated seagrass. Bioscience 71: 1171-1178
- ↑ 17.0 17.1 17.2 17.3 17.4 van Tussenbroek, B.I., Vonk, J.A., Stapel, J., Erftemeijer, P.L.A., Middelburg, J.J. and Zieman. J.C. 2007. Ch. 18. The Biology of Thalassia: Paradigms and Recent Advances in Research. In Larkum, A.W.D. and Orth, R.J. (eds.) Seagrasses: Biology, Ecology and Conservation, pp. 409–439. Springer
- ↑ Brouns, J.J.W.M. 1985. A comparison of the annual production and biomass in three monospecific stands of the seagrass Thalassia hemprichii (Ehrenb.) Aschers. Aquat. Bot. 23: 149–175
- ↑ 19.0 19.1 Photo credit Eric Coppejans - http://www.vliz.be/imis/imis.php?module=person&persid=134
- ↑ Larkum, A.W.D., Drew, E.A. and Ralph, P.J. 2006. Ch. 14 Photosynthesis and Metabolism in Seagrasses at the Cellular Level. In Larkum, A.W.D. and Orth, R.J. (eds.) Seagrasses: Biology, Ecology and Conservation, pp. 409–439. Springer
- ↑ Olsen, J.L. etal. 2016. The genome of the seagrass Zostera marina reveals angiosperm adaptation to the sea. Nature 530: 331
- ↑ Karleskint G. 1998. Introduction to marine biology. Harcourt Brace College Publishers. p.378
- ↑ Zabarte-Maeztu, I., Matheson, F.E., Manley-Harris, M., Davies-Colley, R. and Hawes, I. 2021. Fine sediment effects on seagrasses: A global review, quantitative synthesis and multi-stressor model. Marine Environmental Research 171, 105480
- ↑ Navarro-Mayoral, S., Tuya, F., Prado, P., Marco-Mendez, C., Fernandez-Gonzalez, V., Fernandez-Torquemada, Y., Espino, F., de la Ossa, J.A., Vilella, D.M., Machado, M. and Martínez-Crego, B. 2023. Drivers of variation in seagrass-associated amphipods across biogeographical areas. Marine Environmental Research 186, 105918
- ↑ Levinton J.S. 1995. Marine biology: function, biodiversity, ecology. Oxford University Press. p. 420
- ↑ 26.0 26.1 26.2 United Nations Environment Programme, 2020. Out of the blue: The value of seagrasses to the environment and to people. UNEP, Nairobi.
- ↑ McDevitt-Irwin, J.M., Iacarella, J.C. and Baum, J.K. 2016. Reassessing the nursery role of seagrass habitats from temperate to tropical regions: a meta-analysis. Marine ecology progress series 557: 133–143
- ↑ Duarte, C.M., Marba, N., Gacia, E., Fourqurean, J.W., Beggins, J., Barron, C. and Apostolaki, E.T. 2010. Seagrass community metabolism: assessing the carbon sink capacity of seagrass meadows. Global Biogeochem. Cycles 24 (4)
- ↑ Carr, J., D’Odorico, P., McGlathery, K. and Wiberg, P. 2010. Stability and bistability of seagrass ecosystems in shallow coastal lagoons: role of feedbacks with sediment resuspension and light attenuation. J. Geophys. Res.: Biogeosciences 115, G03011
- ↑ Lafratta, A., Serrano, O., Masqué, P., Fernandes, M., Gaylard, S. and Lavery, P.S. 2023. Seagrass soils sequester up to half the metal emissions of one of the world's largest smelters. Marine Pollution Bulletin 197, 115684
- ↑ Buapet, P., Mohammadi, N.S., Pernice, M., Kumar, M., Kuzhiumparambil, U. and Ralph, P.J. 2019. Excess copper promotes photoinhibition and modulates the expression of antioxidant-related genes in Zostera muelleri. Aquat. Toxicol. 207: 91–100
- ↑ Zhu, Q., Wiberg, P. L. and Reidenbach, M. A. 2021. Quantifying seasonal seagrass effects on flow and sediment dynamics in a back-barrier bay. Journal of Geophysical Research: Oceans, 126, e2020JC016547
- ↑ Lamb, J. B., van de Water, J. A. J. M., Bourne, D. G., Altier, C., Hein, M. Y., Fiorenza, E. A., Abu, N., Jompa, J., and Harvell, C. D. 2017. Seagrass ecosystems reduce exposure to bacterial pathogens of humans, fishes, and invertebrates. Science 355: 731–733
- ↑ Nordlund, L.M., Koch, E.W., Barbier, E.B. and Creed, J.C. 2016. Seagrass ecosystem services and their variability across genera and geographical regions. PLoS One 11 (10), e0163091
- ↑ 35.0 35.1 35.2 Dunic, J.C., Brown, C.J., Connolly, R.M., Turschwell, M.P. and Cote, I.M. 2021. Long-term declines and recovery of meadow area across the world’s seagrass bioregions. Glob Change Biol 27: 4096–4109
- ↑ Moore, K. A. and Jarvis, J. C. (2008). Environmental Factors Affecting Recent Summertime Eelgrass Diebacks in the Lower Chesapeake Bay: Implications for Long-Term Persistence. J. Coast. Res. 55, 135–147
- ↑ Plaisted, H.K., Shields, E.C., Novak, A.B., Peck, C.P., Schenck, F., Carr, J., Duffy, P.A., Evans, N.T., Fox, S.E., Heck, S.M., Hudson, R., Mattera, T., Moore, K.A., Neikirk, B., Parrish, D.B., Peterson, B.J., Short, F.T. and Tinoco, A.I. 2022. Influence of Rising Water Temperature on the Temperate Seagrass Species Eelgrass (Zostera marina L.) in the Northeast USA. Front. Mar. Sci. 9, 920699
- ↑ Oprandi, A., Mucerino, L., De Leo, F., Bianchi, C.N., Morri, C., Azzola, A., Benelli, F., Besio, G., Ferrari, M. and Montefalcone, M. 2020. Effects of severe storm on seagrass meadows. Sci. Total Environ. 748, 141373
- ↑ Alexandre, A., Collado-Vides, L. and Santos, R. 2021. The takeover of Thalassia testudinum by Anadyomene sp. at Biscayne Bay, USA, cannot be simply explained by competition for nitrogen and phosphorous. Marine Pollution Bulletin 167, 112326
- ↑ Zribi, I., Ellouzi, H., Mnasri, I., Abdelkader, N., Ben Hmida, A., Dorai, S., Debez, A., Charfi-Cheikhrouha, F. and Zakhama-Sraieb, R. 2023. Effect of shading imposed by the algae Chaeotomorpha linum loads on structure, morphology and physiology of the seagrass Cymodocea nodosa. Marine Environmental Research 188, 106001
- ↑ 41.0 41.1 van der Heide, T., van Nes, E.H., Geerling, G.W., Smolders, A.J.P., Bouma, T.J. and. van Katwijk, M. M. 2007. Positive feedbacks in seagrass ecosystems: Implications for success in conservation and restoration. Ecosystems 10: 1311–1322
- ↑ Zabarte-Maeztu, I., Matheson, F.E., Manley-Harris, M., Davies-Colley, R.J., Oliver, M. and Hawes, I. 2015. Effects of Fine Sediment on Seagrass Meadows: A Case Study of Zostera muelleri in Pauatahanui Inlet, New Zealand. J. Mar. Sci. Eng. 8, 645
- ↑ Brodersen, K.E., Hammer, K.J., Schrameyer, V., Floytrup, A., Rasheed, M.A., Ralph, P.J., Kühl, M. and Pedersen, O. 2017. Sediment Resuspension and Deposition on Seagrass Leaves Impedes Internal Plant Aeration and Promotes Phytotoxic H2S Intrusion. Front. Plant Sci. 8, 657
- ↑ Suykerbuyk, W., Bouma, T.J., Govers, L.L., Giesen, K., De Jong, D.J., Herman, P., Hendriks, A.J. and Van Katwijk, M.M. 2015. Surviving in Changing Seascapes: Sediment Dynamics as Bottleneck for Long-Term Seagrass Presence. Ecosystems 19: 296–310
Please note that others may also have edited the contents of this article.
|